Airway Management Devices Market Size, Share & Industry Analysis, By Type (Infraglottic Airway Management Devices [Endotracheal Tubes and Tracheostomy Tubes], Supraglottic Airway Management Devices [Oropharyngeal Airways (OPAS), Laryngeal Mask Airways (LMAS), and Nasopharyngeal Airways], Laryngoscopes, and Others), By Application (Anesthesia, Emergency Medicine, and Others), By End User (Hospitals & ASCs, Homecare, and Others), and Regional Forecasts, 2026-2034
Airway Management Devices Market Analysis
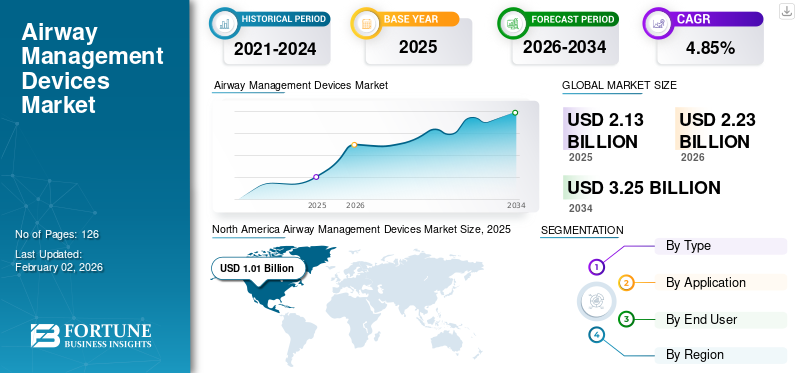
The global airway management devices market size was valued at USD 2.13 billion in 2025 and is projected to grow from USD 2.23 billion in 2026 to USD 3.25 billion by 2034, exhibiting a CAGR of 4.85% during the forecast period. North America dominated the airway management devices market with a market share of 47.1% in 2025.
Airway management devices, such as bag-valve masks, endotracheal tubes, laryngoscopes, and others, provide ventilation and restore breathing in individuals suffering from airway obstruction. Patients suffering from diseases such as cancer, chronic obstructive pulmonary disease (COPD), chronic lung diseases, and others can require effective airway management. Therefore, the growing prevalence of these chronic conditions has been fueling demand for airway management devices.
- According to an article published by the National Center for Biotechnology Information (NCBI) in March 2022, the prevalence of chronic obstructive pulmonary disease (COPD) is around 10.1% among the population aged 40 years and above globally.
- According to data revealed by the American Lung Association, in 2020, 12.5 million people, or 5.0% of adults, reported a diagnosis of COPD, chronic bronchitis, or emphysema. Moreover, as per similar estimates, 148,512 deaths by COPD were reported in the U.S.
Furthermore, the growing prevalence of chronic diseases among the population has emphasized emergency and intensive care admission globally for better treatment of adverse conditions. Thus, the number of patients visiting hospitals in emergency care units has also increased due to the need for timely treatment.
- According to the Centers for Disease Control and Prevention (CDC), in 2022, 16.2 million emergency department visits resulted in hospital admissions in the U.S. and 2.3 million emergency department visits in admission to critical care.
Such an increase in emergency and inpatient admissions of chronic disorders led to a rise in surgical procedures across the globe, further boosting the demand and usage of airway management devices in healthcare centers. Also, the high use of airway devices with the increasing number of anaesthetic procedures for surgical interventions in patients globally is expected to drive the growth of the airway management devices market.
Download Free sample to learn more about this report.
Global Airway Management Devices Market Overview
Airway Management Devices Market Size:
- 2025 Value: USD 2.13 Billion
- 2026 Value: USD 2.23 Billion
- 2034 Forecast Value: USD 3.25 Billion, with a CAGR of 4.85% from 2026–2034
Airway Management Devices Market Share:
- North America led the airway management devices market with a 47.1% share in 2025, driven by high R&D spending and new product launches.
- By type, the infraglottic airway management devices segment is expected to hold a 55.6% share in 2025.
Key Country Highlights:
- The airway management devices market in Japan is expected to reach USD 0.01 billion by 2026.
- China is forecast to witness a strong CAGR of 3.10%, while Europe is anticipated to grow at a CAGR of 5.6% during the forecast period.
- By application, the anesthesia segment is projected to generate USD 1,261.3 million in revenue by 2025.
During the COVID-19 pandemic in 2020, the market experienced a decline in its growth due to the limited number of patients visiting hospitals and clinics. Moreover, the limited availability of resources due to lockdown restrictions also negatively impacted the market. However, the pending elective surgeries rebounded quickly after the initial COVID-19 shutdowns, and inpatient volumes increased across the globe. These led to the increased demand and usage of these devices in anaesthetic and surgical procedures, further propelling the market growth in 2021.
Airway Management Devices Market Trends
Adoption of Airway Management Devices has been Increasing Significantly Among Neonates
The number of premature births has been increasing significantly across the globe due to certain risk factors such as infections, changes in lifestyle activities, increasing stress in pregnant women, and others.
- For instance, according to data published by the World Health Organization (WHO) in 2023, there were around 13.4 million premature births worldwide in 2020, which accounts for more than one premature birth out of 10.
- Similarly, according to data published by the Centers for Disease Control and Prevention (CDC), in 2022, one out of 10 births in the U.S. was preterm. Moreover, the preterm birth rate increased by 4.0% from 2020 to 2021 in the U.S.
With the increasing number of preterm births, the incidence of breathing disorders in these births has also been rising significantly. This has been fueling the demand for airway management in neonates. Moreover, the primary key companies across the globe are introducing these devices with advanced technologies for pediatric patients suffering from breathing disorders, further driving the market growth.
- According to an article published by Rapid Medtech Communications Ltd. in May 2021, globally, 415,000 neonates and infants require invasive mechanical ventilation per year and are intubated for greater than 24 hours. Moreover, approximately 75,000 neonates and infants experience at least one unplanned extubation.
- In May 2021, Medtronic plc announced the launch of the SonarMed airway monitoring system for pediatric patients in the U.S. The device is designed to check for obstructions in the endotracheal tube and verify positions in real time, helping clinicians diagnose neonatal more efficiently. Adopting the SonarMed airway monitoring system can help reduce the frequency of events such as intubation or extubation, which can help improve outcomes and reduce healthcare costs.
Therefore, the rising incidence of pre-term births and respiratory disorders has increased the demand for airway management in neonates, fueling the market growth.
Download Free sample to learn more about this report.
Airway Management Devices Market Growth Factors
Rising Number of Joint Ventures and Start-ups Among Industry Players to Fuel the Market Growth
The growing burden of respiratory disorders across the globe has been fueling the demand for airway medical devices. In order to fulfil this demand, market players have increased their focus on expanding their production capacity for the development and launch of new advanced products.
- For instance, in April 2021, Medtronic launched the largest R&D center in India to launch new devices, advancing new technologies in surgical navigation and imaging, airway devices, ventilators, and many others.
- In August 2020, the Food and Drug Administration (FDA) approved the use of LifeMech A-VS, a low-cost emergency resuscitator developed by LifeMech, Inc., that was designed to support the breathing management of patients during the COVID-19 pandemic.
Furthermore, various players are adopting strategies like joint ventures and start-ups to develop novel devices with expansive product portfolios for patients suffering from breathing disorders. Also, the rising focus of key players on developing technologically advanced devices in airway management to introduce a range of suitable products for both adults and neonates will boost the market during the forecast period.
- In January 2022, ICU Medical Inc., completed the acquisition of Smiths Medical from Smiths Group PLC, to increase the product portfolio globally in devices for airway management, vital care products, ambulatory infusion devices, and others to enhance the patient treatment experience and improve overall outcomes.
Therefore, such high investments by medical device manufacturers in R&D centers and for the production of devices are anticipated to fuel the introduction of advanced devices across various emerging nations, further propelling the airway management devices market growth.
RESTRAINING FACTORS
Complications Associated With the Use of Airway Devices to Limit the Adoption
The demand for effective treatment options to treat respiratory disorders is increasing globally. Intubation using airway management devices is often a life-saving procedure. Still, long-term use of these devices may pose short or long-term severe risks and potential complications such as regurgitation and aspiration of gastric contents, compression of vascular structures, trauma, and nerve injury. The incidence of such complications, including trauma and nerve injury, is comparatively low, but using some medical devices may carry significant risks.
- For instance, as per data published by the National Center for Biotechnology Information (NCBI) in 2021, the most common complication associated with a tracheostomy is bleeding, which had an estimated incidence of 0.6%-5.0% globally.
- Furthermore, as per another research study published by the National Center for Biotechnology Information in 2023, the incidence of complications such as cardiovascular instability, hypoxaemia, and cardiac arrest due to the use of airway management devices was 42.6%, 9.3%, and 3.1%, respectively.
Laryngeal mask airway devices can also pose a few complications in pediatric patients, including soft-tissue trauma, vomiting, regurgitation, stridor, and airway restriction on misplacement of the device. Also, potential long-term complications of using laryngeal mask airway devices, including laryngospasm, and venous and capillary congestion, are rising across the globe.
- According to an article by Cureus in April 2022, a trial was conducted on 106 randomized anesthesia patients of the American Society of Anaesthesiology using a laryngeal mask airway. The complications results were that about 6.6% had laryngospasm, 20.0% had airway obstruction, 15.0% had a cough, and 10.0% of patients had observed peripheral oxygen desaturation. The trial concluded that adverse respiratory events happen in awake and deep planes of anesthesia after removing the laryngeal mask airway device in children.
Thus, the rising number of severe risks and complications reported in patients by use of these devices are expected to hamper their extensive adoption, further restraining the market growth.
Airway Management Devices Market Segmentation Analysis
By Type Analysis
Increasing Launch of Technologically Advanced Devices to Augment Segment Growth for Infraglottic Airway Management Devices
The airway management devices market is segmented by type into infraglottic airway management devices, supraglottic airway management devices, laryngoscopes, and others. Among types, the infraglottic airway management devices segment accounted for the largest market share in 2023. The dominance was attributed to the rise in the prevalence of chronic respiratory disorders globally and the rising adoption of these devices by healthcare providers in intensive care units and operating rooms for anaesthetic and emergency procedures. By type, the infraglottic airway management devices segment is expected to hold a 55.46% share in 2026.
- For instance, according to an article published by BJA Education in 2019, more than 12,000 tracheostomy devices are inserted annually by physicians in the U.K., and up to 20.0% of patients in ICU are managed with a tracheostomy.
Moreover, the increasing focus of several key players on introducing technologically advanced devices and adding new widgets to their existing airway management product portfolio to provide enhanced patient care is primarily contributing to the segment's growth.
- In May 2021, Ambu A/S obtained U.S. FDA clearance for the Ambu VivaSight 2 DLT, the latest innovation in airway tubes for safe and cost-effective use by clinicians with a built-in camera used during single-lung ventilation procedures.
- In January 2020, Smiths Medical announced adding a PVC-based tracheostomy tube portfolio to its Portex Tracheostomy Tubes and Kits, designed to help clinicians provide the best patient care.
On the other hand, the supraglottic airway management devices segment is expected to reach a comparatively higher CAGR during the forecast period. The segment's growth is attributed to increasing investments in production and the launch of single-use devices of airway management by key companies and high demand of these devices among healthcare professionals is likely contribute to the market growth in the region.
- In March 2020, Intersurgical Ltd introduced the newly advanced i-gel, a single-use supraglottic airway device used during spontaneous or intermittent positive pressure ventilation (IPPV) of the unconscious patient.
To know how our report can help streamline your business, Speak to Analyst
By Application Analysis
Increasing Number of Anesthesia Procedures will Augment the Segment Growth
Based on application, the market is segmented into anesthesia, emergency medicine, and others.
By application, the anesthesia segment accounted for the highest airway management devices market share of 59.07% in 2026. Anesthesia is used during the surgical procedures to provide sedation. The increasing number of surgical procedures globally is responsible for the segment’s dominance. By application, the anesthesia segment is projected to generate USD 1,261.3 million in revenue by 2025.
- According to an article published by Springer Nature in September 2020, 11.0% of the global burden of disease requires surgical care, anesthesia management, or both. Some studies have estimated this burden to be as high as 30.0%.
On the other hand, the emergency medicine segment is projected to register a comparatively higher CAGR owing to its rising emergency visits in hospitals & ASCs across developed countries and increasing prevalence of trauma and accidental injuries among the population.
- As per European Society of Emergency Medicine (ESEM) estimates published in July 2020, emergency department visit rate is 100-300 visits per 1,000 habitants per year in Europe. Moreover, as per similar estimates, 25.0% of the European population visit the emergency departments annually.
By End User Analysis
Surge in Surgical Procedures in Hospitals & ASCs to Propel Growth
Based on end user, the market is segmented into hospitals & ASCs, homecare, and others.
Hospitals & ASCs segment accounted for the highest market share of 78.56% in 2026. The highest share of the segment was attributed to the rising number of hospital visits, especially in emerging countries.
- According to data published by BioMed Central Ltd, in 2021, around 446,484 emergency department admissions were recorded in Singapore hospitals in a period of 10 years.
The homecare segment is expected to grow at a considerable CAGR during the forecast period. The growth is attributable to the rising number of the global geriatric population with chronic conditions across globe and high adoption of homecare settings amongst the geriatric population.
- According to an article published by Definitive healthcare in 2021, before the pandemic, 5.0 million people in 2019 received care at home in the U.S. However, post-COVID-19 pandemic, this number increased to 12.0 million by the end of 2020.
REGIONAL INSIGHTS
Rise in the Introduction of Technologically Advanced Airway devices to Augment North America Market
The market can be segmented by region into North America, Europe, Asia Pacific, Latin America, the Middle East, and Africa.
North America Airway Management Devices Market Size, 2025 (USD Billion)
To get more information on the regional analysis of this market, Download Free sample
North America
The North America region captured 47.10% of the global market in 2025, generating USD 1.01 billion in revenue, and is projected to reach USD 1.05 billion in 2026. Key players' rising R&D and introduction of new technologies in airway management devices to help aid in patient treatment are likely to drive the North American market's growth. Moreover, increasing collaborations among manufacturing players and rising approvals of new airway devices with extensive presence across the region further boosted the North American market. The U.S. market is projected to reach USD 0.84 billion by 2026.
- For instance, in May 2021, Olympus Corporation announced the FDA clearance of its Airway Mobilescopes, the MAF-TM2, MAF-GM2, and MAF-DM2, enabling providers to perform a variety of upper and lower airway management procedures.
Europe
Europe maintained a strong presence in the global market, reaching USD 0.67 billion in 2025, accounting for 31.60% share, and is expected to reach USD 0.71 billion in 2026. Europe is the second most dominated region in terms of revenue and market share. The European market held a significant share due to the increasing number of emergency hospital visits and inpatient admissions in hospitals, owing to the high prevalence of chronic disorders among the population. Similarly, increased demand for these devices for the treatment of such a high number of disorders will further propel the market growth in Europe. The UK market is projected to reach USD 0.05 billion by 2026, and the Germany market is projected to reach USD 0.19 billion by 2026.
- Europe is anticipated to grow at a CAGR of 5.6% during the forecast period.
- According to an article published by the Interactive Health Atlas of Lung Conditions in England (INHALE) in February 2022, the emergency hospital admissions rate in England for respiratory disease has steadily and significantly increased from the financial year 2014, with a rate of 1,228.0 per 0.1 million of the population, to a rate of 1,592.0 per 0.1 million in the financial year 2020.
Asia Pacific
In 2025, Asia Pacific generated USD 0.3 billion, contributing 14.10% to global market revenue, and is projected to grow to USD 0.31 billion in 2026. The market share in Asia Pacific region is driven by the rising prevalence of respiratory disorders amongst the Asia Pacific population, and the increasing demand for emergency care owing to high disease prevalence is anticipated for the region's growth at the highest CAGR. The Japan market is projected to reach USD 0.01 billion by 2026, the China market is projected to reach USD 0.05 billion by 2026, and the India market is projected to reach USD 0.07 billion by 2026.
- According to an article published by NCBI in July 2021, the prevalence of COPD in Asia Pacific countries is estimated at 14.5% in Australia, 16.7% in China, and 8.6% in Japan.
- The airway management devices market in Japan is expected to reach USD 5.5 million by 2025.
- China is projected to witness a strong CAGR of 3.10% during the forecast period.
Latin America
The Latin America market generated USD 0.1 billion in 2025, representing 4.70% of the global market landscape, and is expected to reach USD 0.1 billion in 2026. On the other side, the Latin American market is likely to grow slower. The growth in the region is owing to rising anesthetic procedures amongst the population and high demand for devices of airway management across hospitals and homecare settings. The Middle East & Africa region is expected to grow at a moderate CAGR owing to the rising number of hospitals and homecare centers providing treatment in the region, creating demand for airway devices during the forecast period.
Middle East & Africa
Middle East & Africa recorded a market size of USD 0.05 billion in 2025, capturing 2.50% of the global market share, and is projected to reach USD 0.06 billion in 2026.
List of Key Companies in Airway Management Devices Market
Robust Investments for New R&D Centers by Key Players to Propel Market Growth
The airway management devices industry is primarily dominated by manufacturers such as Smiths Medical (ICU Medical), Teleflex Incorporated, and other prominent players. The top market players are focusing on accelerating investments in medical device production centers and rising collaborations & acquisitions owing to the high demand for these devices across the globe. These factors are anticipated to augment the airway devices market growth.
- In May 2021, Teleflex Incorporated and Medline Industries, Inc. signed a definitive agreement to acquire Teleflex Incorporated’s non-invasive ventilation products (ventilation support through the patient's upper airway using a mask or similar device) and others that will further boost the company’s financial flexibility for its growth strategy.
Similarly, other prominent players in the market, including Ambu A/S, and Convatec plc, are continuously engaged in key developments, such as enhancing their distribution channel across the global market through strategic partnerships and extensive product approvals across the globe. Such initiatives by key players are projected to propel the global market and register growth by 2032.
- In May 2021, Ambu A/S obtained U.S. FDA clearance for the Ambu VivaSight 2 DLT, the latest innovation in airwsay tubes for safe and cost-effective use by clinicians with a built-in camera used during single-lung ventilation procedures.
Thus, such continuous engagement in the production of medical devices by several market players and a rising focus on enhancing their distribution channel across the global market through R&D investments in order to establish their footprints in emerging nations will further propel the market.
LIST OF KEY COMPANIES PROFILED:
- Medtronic (Ireland)
- Teleflex Incorporated (U.S.)
- KARL STORZ SE & Co. KG (Germany)
- ICU Medical (U.S.)
- Convatec Inc. (U.K.)
- Ambu A/S (Denmark)
- Intersurgical Ltd (U.K.)
- VBM Medizintechnik GmbH (Germany)
- VYAIRE MEDICAL, INC. (U.S.)
- Flexicare (Group) Limited (U.K.)
KEY INDUSTRY DEVELOPMENTS:
- June 2023 – Airway Management expanded its airway management devices product portfolio with the launch of Morf Nasal Mask.
- January 2022 - Intersurgical Ltd announced the acquisition of Pulmodyne, Inc., a U.S., and Malaysian-based manufacturer, complementing in providing high-quality innovative airway and respiratory products for use in prehospital, emergency care, critical care, and at home. The acquisition will further help to provide Intersurgical with an elaborative presence in the U.S. market and for increased sales of the Pulmodyne ranges through the extensive global Intersurgical Ltd network.
- May 2021 - Teleflex Incorporated and Medline Industries, Inc., signed a definitive agreement to acquire Teleflex Incorporated non-invasive ventilation products (ventilation support through the patient's upper airway using a mask or similar device) and others that will further boost the company’s financial flexibility for its growth strategy.
- March 2021 - Convatec Group PLC announced the acquisition of Cure Medical, LLC bringing together the two innovative manufacturers of continence care products, to offer a more comprehensive range of products & services management of chronic conditions.
- December 2020 - Flexicare Medical Limited along with Medisize B.V., enhanced their presence in Europe region supported by the sales and distribution networks in European geographies including Netherlands, Czech Republic, and others in airway management and anesthesia devices.
REPORT COVERAGE
The global airway management devices market report provides detailed market analysis and focuses on crucial aspects such as leading players, product types, and major indications of the product. Additionally, it offers insights into market trends, key industry developments such as mergers, partnerships, & acquisitions, and the impact of COVID-19 on the market. In addition to the factors mentioned above, the report includes the factors that have contributed to the market's growth in recent years with a regional analysis of different segments.
Request for Customization to gain extensive market insights.
Report Scope & Segmentation
|
ATTRIBUTE |
DETAILS |
|
Study Period |
2021-2034 |
|
Base Year |
2025 |
|
Forecast Period |
2026-2034 |
|
Historical Period |
2021-2024 |
|
Growth Rate |
CAGR of 4.85% from 2026-2034 |
|
Unit |
Value (USD billion) |
|
Segmentation |
By Type
|
|
By Application
|
|
|
By End User
|
|
|
By Region
|
Frequently Asked Questions
According to Fortune Business Insights, the global airway management devices market was valued at USD 2.13 billion in 2025, with projections reaching USD 3.25 billion by 2034.
Registering a CAGR of 4.85%, the market will exhibit steady growth in the forecast period.
North America remains the leading region due to its advanced healthcare infrastructure, high spending on surgical care, and rapid adoption of technologically advanced airway management systems.
Growth is driven by rising surgical procedures, increased ICU admissions, growing prevalence of respiratory diseases like COPD and asthma, and greater adoption of emergency airway tools in trauma centers.
Leading manufacturers include Medtronic, Teleflex, Smiths Medical, Ambu A/S, and Vyaire Medical. These players focus on innovation in airway devices, expanding ICU solutions, and launching portable technologies for emergency and anesthesia applications.
Key trends include the rise of video laryngoscopes, single-use supraglottic devices, AI-integrated airway visualization tools, and increased demand for pediatric airway management solutions.
High-demand segments include infraglottic devices (e.g., endotracheal tubes), supraglottic airway devices, and resuscitators. Hospitals are also rapidly transitioning to disposable airway devices to reduce infection risk.
Emergency care and critical care units increasingly rely on real-time airway access and intubation tools. Demand is particularly strong for portable, easy-to-use devices that support rapid response teams.
Emerging countries are investing in surgical and critical care infrastructure. Rising health awareness, better neonatal and trauma care access, and government funding in Asia-Pacific and Latin America present major opportunities.
Get 20% Free Customization
Expand Regional and Country Coverage, Segments Analysis, Company Profiles, Competitive Benchmarking, and End-user Insights.
Related Reports
-
US +1 833 909 2966 ( Toll Free )
-
Get In Touch With Us