Spinal Muscular Atrophy (SMA) Treatment Market Size, Share & Industry Analysis, By Product (Nusinersen and Onasemnogen Abeparvovec), By Disease Type (Type 1 SMA, Type 2 SMA and Others), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies and Others), and Regional Forecast, 2026-2034
Spinal Muscular Atrophy Treatment Market Size and Industry Overview
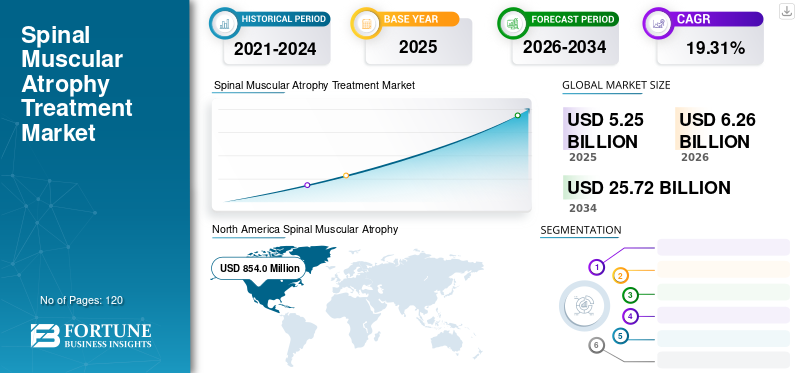
The global spinal muscular atrophy treatment market size was valued at USD 5.25 billion in 2025. The market is projected to grow from USD 6.26 billion in 2026 to USD 25.72 billion by 2034, exhibiting a CAGR of 19.31% during the forecast period. North America dominated the global market with a share of 49.65% in 2025. Growing incidence of spinal muscular atrophy across the globe is expected to boost the demand for advanced therapies to reduce the disease burden.
The global market growth is driven by a significant increase in the number of product launches despite the relative rarity of the disorder and high costs associated with the approved drugs. The treatment of spinal muscular atrophy through the gene therapy drug called Zolgensma by the Swiss drugmaker Novartis comes with a price tag of USD 2.1 million, for a one-time treatment. Despite such high costs, the severity of the disorder, which is considered the leading genetic cause of death in infants, is driving the adoption of these expensive drugs. According to an article published by ScienceDaily, an estimated 1 in 11,000 babies globally are born with spinal muscular atrophy and before the approval of the currently used therapeutics such as Spinraza and Zolgensma, most of the children with Type 1 disorder died before the age of 2.
Download Free sample to learn more about this report.
Spinal Muscular Atrophy Treatment Market Snapshot & Highlights
Market Size & Forecast:
- 2025 Value: USD 5.25 billion
- 2026 Value: USD 6.26 billion
- 2034 Forecast Value: USD 25.72 billion
- CAGR: 19.31% from 2026–2034
Market Share:
- North America dominated the global market with a 49.65% share in 2018, driven by early treatment approvals, strong reimbursement frameworks, and higher treatment adoption rates.
- By product, Nusinersen held the largest share in 2018 as it was the only approved treatment at the time. However, Onasemnogen Abeparvovec is projected to gain strong momentum post-2019 due to global market launches and increasing patient adoption.
Key Country Highlights:
- Japan: Government-backed reimbursement access for all patients boosts treatment uptake.
- United States: Rapid approvals, high awareness, and private insurance coverage fuel adoption despite high costs (Zolgensma at USD 2.1M and Spinraza at USD 750K first year).
- China: Reimbursement approval processes underway; large patient base expected to drive future growth.
- Europe: Countries like Austria, France, Germany, and Italy offer full reimbursement, supporting market expansion.
Spinal Muscular Atrophy Treatment Market Trends
Increasing R&D Initiatives in Treatment of Spinal Muscular Atrophy and Presence in Pipelines of Key Players
One of the key market trends in the spinal muscular atrophy treatment market is the increasing R&D initiatives resulting in the appearance of potential treatment options in the pipelines of major companies. Some of the major companies with pipeline candidates in the advanced stages of clinical trials include Genentech/Roche’s pipeline candidate of Risdiplam, which recently received a priority review from the FDA and is expected to receive a decision on approval from the FDA by May 2020. Such R&D initiatives are leading to the creation of greater awareness and choices of treatment for the patients. This is estimated to propel the number of patients opting for treatment for the patients in the coming years. This trend is following the recent trend of greater R&D for rare diseases.
Download Free sample to learn more about this report.
Increasing Reimbursement Access to Patients Across the Globe
One of the market trends in the global spinal muscular atrophy (SMA) treatment market is increasing access to the reimbursement of expensive drugs across the world in many regions. This is anticipated to positively affect the adoption of the expensive gene therapeutics, leading to a substantial growth of the global market. Some of the countries that have granted reimbursed access to all patients include Austria, France, Germany, Italy in Europe while Israel in the Middle East and Hong Kong in Asia have granted the all patients, reimbursed access. In the United States, the reimbursed access will be as approved by individual insurance providers and those patients rejected may approach Biogen’s humanitarian program
MARKET DRIVERS
Growing Incidence of the Disorder to Augment the Market
The overall burden of spinal muscular atrophy is one of the highest in terms of cost and healthcare services in the rare disorder. The rising incidence of spinal muscular atrophy due to the increasing population is one of the key factor for the growth in global SMA treatment market. Despite being considered a rare disorder, according to Spinal Muscular Atrophy Foundation it is estimated that 1 in every 6,000 to 10,000 babies born worldwide are diagnosed with the disorder. Thus, the rising prevalence of the disorder is one of the key factors for the increasing revenue generation from market. the increasing product launches and rising awareness regarding novel therapies are some of the major factor influencing the growth of the market. the awareness about the lethal repercussions of the disorder and surge in the demand for quality treatment options will significantly aid in the development of the market .
New Product Launches with High Efficacies to Drive the Global Market
The increasing incidence and awareness for spinal muscular atrophy have increased remarkably which is propelling the demand for efficient therapeutic options leading to better patient outcomes. In recent times, there have been two substantial and very significant product launches in gene therapy for the treatment of spinal muscular atrophy. Spinal muscular atrophy was a rare disorder for a very long time which had no treatment options which often proved fatal to the infants and children suffering from the disorders. Two product launches: Spinraza (Nusinersen) by Biogen and Zolgensma (Onasemnogene abeparvovec) by Novartis for the treatment of the disorder are anticipated to drive the market greatly. Coupled with high efficiency of the therapeutics, these factors are anticipated to drive market.
MARKET RESTRAINT
High Costs Associated with the Approved Products To Restrict Spinal Muscular Atrophy Treatment Market Growth
One of the key factors that have limited the growth of the global market are the high prices associated with the treatment of rare gentic diseases. For instance; Spinraza is estimated to cost US$ 750,000 for the first year, US$ 375,000 for every year thereafter, bringing the costs of treatment using this therapy over a decade to US$ 4.1 million. While Novartis’ treatment of Zolgensma is estimated to cost US$ 2.1 million for a one-time treatment. These high costs often affect the adoption of treatment for many patients, but an overwhelmingly large number of countries across the world have favourable reimbursement policies. Steps as such are predicted to limit the adoption of high cost products.
Segmentation Analysis
By Product Analysis
To know how our report can help streamline your business, Speak to Analyst
Onasemnogen Abeparvovec to Gain Momentum during the Forecast Period
Based on product, the market is segmented into Nusinersen and Onasemnogen Abeparvovec. Nusinersen accounted for maximum share in 2018, which is attributable to the fact that in 2018, Nusinersen was the only approved product available in the market. However, Onasemnogen Abeparvovec who gained approval in 2019, is anticipated to be launched in several markets over the periods of 2019 and 2020. Onasemnogen Abeparvovec is expected to witness growth in the coming years owing to the rising launches in the number of markets across the globe and greater patient adoption.
By Spinal Muscular Atrophy (SMA) Disease Type Analysis
Type 1 SMA to Account for Maximum Share
By disease type, the global spinal muscular atrophy treatment market is categorized into Type 1 SMA, Type 2 SMA and Others. Type 1 SMA generated the highest revenue amongst the three categories in 2018 and is expected to remain dominant in terms of revenue throughout the forecast duration. Type 1 SMA is anticipated to remain the most dominant because the majority of the cases of the disorder are of this type, which has led to it dominating in terms of market share. Increasing awareness of the Type 2 SMA and increasing adoption of treatment is also anticipated to drive the growth of this type in the forecast period.
By Distribution Channel Analysis
Spinal Muscular Atrophy (SMA) Treatment Centers And Hospitals To Retain The Leading Position
By distribution channel, the global SMA treatment market is categorized into hospital pharmacies, retail pharmacies, and others. In terms of value, hospital pharmacies accounted for the leading market share in 2018, which is attributable to the fact that the majority of the medications for treatment can be only administered intravenously and can so only be done by trained medical professionals. Hence, the retail pharmacies segment registers a comparatively lower CAGR.
REGIONAL ANALYSIS
SMA treatment market in North America was valued at USD 854.0 million in 2018. Early approvals for SMA treatment products in the U.S., coupled with adequate reimbursement, has led to growing number of patients being treated in 2018. Europe is expected to hold the second leading position in this market by witnessing strong growth in long-term period. As per our market research study, Asia Pacific is expected to witness relatively significant growth in terms of spinal muscular atrophy treatment market value. Funding by public and private players to improve the accessibility of high-cost treatment options is likely to augment the expansion of the market during the forecast period. For instance, in the key regions of Asia such as Japan and Hong Kong have granted the status of reimbursed access to all patients, while in China, the process of reimbursement approval is currently underway. Such a positive reimbursement scenario for spinal muscular atrophy treatment is projected to boost the expansion of the market in Asia Pacific during the forecast period.
North America Spinal Muscular Atrophy (SMA) Treatment Market Size, 2025 (USD Million)
To get more information on the regional analysis of this market, Download Free sample
The rest of the world comprises of the regions of Latin America and Middle East & Africa. In Latin America, the rising incidence of the disorder among the population is boosting the spinal muscular atrophy treatment market growth. Middle East & Africa is estimated to reflect slower-to-moderate growth during the forecast period, which is attributable to the delayed diagnosis of the disorder in African countries. However, positive reimbursement policies in the region in the countries in Middle East and growing awareness for the severity of the disease are the key factors estimated to propel the revenue from Middle East & Africa spinal muscular atrophy (SMA) treatment market during the forecast period.
KEY INDUSTRY PLAYERS
Biogen and Novartis AG focuses on introduction of advanced products to gain strategic position across the globe
Biogen and Novartis AG accounted for the complete market share in 2018, owing to their two main products for the treatment of spinal muscular atrophy. Spinraza (Nusinersen) by Biogen and Zolgensma (Onasemnogene abeparvovec) by Novartis are the only two products approved for the treatment of this disorder. Hence, these two companies hold complete dominance in terms of the market share. The other companies such as Genentech, Inc. (F. Hoffmann-La Roche Ltd) and Astellas Pharma hold key pipeline candidates, some of which are in the advanced stages of clinical trials.
LIST OF KEY COMPANIES PROFILED:
- Biogen
- Novartis AG
- Genentech, Inc./ F. Hoffmann-La Roche Ltd
- Astellas Pharma Inc.
- Genzyme Corporation
- Others
KEY INDUSTRY DEVELOPMENTS:
- May 2019 - Zolgensma (onasemnogene abeparvovec-xioi) by Novartis AG received the approval from the U.S. FDA for the first and only gene therapy for pediatric patients with spinal muscular atrophy (SMA)
- December 2016 – The U.S. FDA approved the first-ever drug for spinal muscular atrophy called Spinraza (Nusinersen) from Biogen
REPORT COVERAGE
The report offers qualitative and quantitative insights on SMA treatment products and the detailed analysis of market size & growth rate for all possible segments in the market. Along with this, the report provides an elaborative analysis of market dynamics, emerging trends, and competitive landscape. Key insights offered in the report are the prevalence of spinal muscular atrophy for key countries, key industry developments, pipeline analysis, new product launches, reimbursement policies, regulatory scenario, and key industry trends.
Report Scope & Segmentation
|
ATTRIBUTE |
DETAILS |
|
Study Period |
2021-2034 |
|
Base Year |
2025 |
|
Forecast Period |
2026-2034 |
|
Historical Period |
2021-2034 |
|
Unit |
Value (USD million) |
|
Segmentation |
By Product
|
|
By Disease Type
|
|
|
By Distribution Channel
|
|
|
By Geography
|
Frequently Asked Questions
As per our (Fortune Business Insights) study, the global market is predicted to reach USD 14.49 Bn by 2026 with a CAGR of 28.9% (2019 -2026)
In 2018, the global market was USD 1.72 Bn, and it is anticipated to reach USD 14.49 Bn by 2026 growing at a stellar CAGR of 28.9% during the forecast period (2019 -2026)
The market is projected to grow at a CAGR of 28.9% during the forecast period (2019-2026).
The market in North America stood at USD 854.0 million in 2018.
Nusinersen among the product is the leading segment, owing to its status as the only available therapeutic in the market as of 2018 and improved treatment outcomes of its patients.
Significant rise in the prevalence of the disease, strong R&D initiatives and new product launches are few of the key factors driving the growth of the global market.
In the global market, some of the key players are Biogen and Novartis AG.
Get 20% Free Customization
Expand Regional and Country Coverage, Segments Analysis, Company Profiles, Competitive Benchmarking, and End-user Insights.
Related Reports
-
US +1 833 909 2966 ( Toll Free )
-
Get In Touch With Us