IV Flush Syringe Market Size, Share & COVID-19 Impact Analysis, By Type (Saline and Heparin), By End User (Hospitals & Ambulatory Surgical Centers, Clinics, and Others), and Regional Forecast, 2026-2034
KEY MARKET INSIGHTS
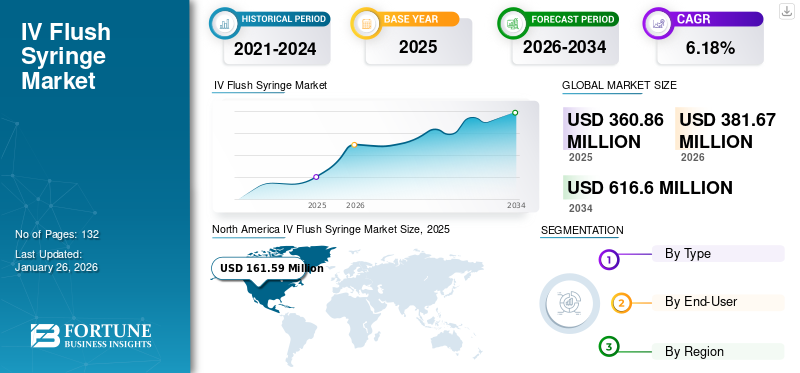
The global IV flush syringe market size was valued at USD 360.86 million in 2025 and is projected to grow from USD 381.67 million in 2026 to USD 616.60 million by 2034, exhibiting a CAGR of 6.18% during the forecast period. North America dominated the IV flush syringe market with a market share of 44.78% in 2025.
Based on our analysis, the global market exhibited a negative growth of 7.4% in 2024 as compared to the average year-on-year growth during 2019-2032. The global impact of COVID-19 has been unprecedented and staggering, with witnessing a negative impact on demand across all regions amid the pandemic. The rise in CAGR is attributable to this market’s growth and demand, returning to pre-pandemic levels once the pandemic is over.
The rapid rise in various chronic conditions, including asthma, diabetes, cancer, and others. This factor is leading to an increasing number of hospital inpatient admissions. For instance, the American Hospital Association conducts an annual survey of hospitals in the U.S., in 2024, the total admissions in all U.S. hospitals were 36.2 million. Increasing hospital admissions leads to the increasing number of patients using IV catheters for the administration of multiple medications. This factor leads to increased demand for flushing IV lines using IV flush syringes in healthcare facilities globally. Additionally, high demand for prefilled syringes is observed among healthcare service providers to prevent cross-contamination, contributing to the demand for prefilled IV flush syringes.
This factor is further augmented by the surging investment by key players in highly advanced production facilities to develop and manufacture new and advanced products. For instance, in December 2020, BD (Becton, Dickinson, and Company) announced the plan of investing around USD 1.2 million over a period of 4 years. This investment will strengthen manufacturing capacity and technology for pre-fillable syringes. Such new facilities and innovative products are projected to contribute to the overall market growth.
Decline in non-COVID-19 Medical Admissions amid COVID-19 Pandemic to Impede Market Growth
The market witnessed negative growth in terms of revenues in 2020, owing to the impact of COVID-19 on several segments. The demand for IV flush syringes decreased in the year 2020 owing to a decline in non-COVID-19 medical admissions. For instance, according to the Federal Statistical Office of Germany, the total number of hospital admissions in Germany reported a decline of 13.2% in 2024. Country lockdowns have caused a considerable decrease in the number of non-COVID-19 related healthcare services and procedures, resulting in a drop in sales of these devices.
Furthermore, elective and non-essential procedures were being delayed in the majority of the countries that the outbreak has impacted to avoid the spread of the infection. For instance, on March 18th, 2020, the Centers for Medicare & Medicaid Services (CMS) announced that all elective and non-essential medical, surgical and dental procedures would be delayed to preserve the healthcare resources for the treatment of COVID-19 patients.
This factor led to a decline in surgical procedures resulting in lower demand for medication delivery products such as syringes, flush syringes, and others. Additionally, the restrictions imposed by the country government due to the COVID-19 pandemic. This factor led to supply chain and logistics disruptions worldwide, resulting in the delay of shipments, which ultimately hinder market's overall growth.
Download Free sample to learn more about this report.
Global IV Flush Syringe Market Snapshot & Highlights
Market Size & Forecast:
- 2025 Market Size: USD 360.86 million
- 2026 Market Size: USD 381.67 million
- 2034 Forecast Market Size: USD 616.60 million
- CAGR: 6.18% from 2026–2034
Market Share:
- North America dominated the IV flush syringe market with a 44.78% share in 2025, driven by the significant adoption of advanced products, rising inpatient admissions, and an increasing number of surgical procedures.
- By type, flush syringes held the largest market share, owing to their high usage in inpatient settings for multiple medication administrations, especially in chronic disease treatments like chemotherapy.
Key Country Highlights:
- United States: Demand is propelled by increasing hospital admissions for chronic diseases and regulatory emphasis on single-use prefilled syringes to prevent cross-contamination.
- Europe: Market growth is driven by rapid healthcare infrastructure development, technological advancements in syringe designs, and rising hospital procedures.
- China: The adoption of prefilled syringes is accelerating due to healthcare providers shifting away from traditional bedside preparation methods in hospitals.
- Japan: The market benefits from the high average length of hospital stays, leading to an increased frequency of IV flush requirements in healthcare facilities.
LATEST TRENDS
Download Free sample to learn more about this report.
Regional Expansion and Development of Innovative Products to offer Growth Opportunity for Key Players
In the majority of developing countries like India, Brazil, and others. IV flushing is carried out using a traditional method which involves bedside preparation of the saline solution and then filling this solution in a syringe and flushing the IV line. Currently, this generic method is adopted by most hospitals and healthcare centers in developing countries, and low awareness limits the adoption of prefilled IV flush syringes in low and middle-income countries. However, established guidelines and protocols by regional and national authorities and market players' effort have led to a shift towards the preference of prefilled IV flush syringes in healthcare settings. Healthcare providers are now shifting from traditional flushing IV lines in inpatient treatment.
Additionally, the market is witnessing the growing demand for easy-to-use, convenient, cost-effective, and safe alternatives for conventional delivery systems. Thus, leading players are expanding their product portfolio to introduce advanced syringe designs, high-quality material, and product launch to fulfill the growing demands of patients and healthcare providers globally.
DRIVING FACTORS
Rise in Awareness and Favorable Guidelines for Single Use Syringes to Boost Product Demand
In terms of the modern demographics, the world's population has experienced a significant increase in several chronic diseases. According to the data from Healthcare Cost & Utilization Project, in the U.S., chronic diseases such as arthritis, diabetes, COPD, and cardiac diseases are responsible for longer inpatient stays in hospital settings in the U.S. Increasing number of admissions in hospitals requiring treatment is fueling the demand for flushing of IV lines.
Additionally, several guidelines are being set up by various regulatory bodies to favor single-use syringes. Regulatory authorities and healthcare departments in various countries are spreading awareness of single-use of syringes among healthcare professionals. Numerous regulatory authorities and healthcare organizations provided guidelines for single-use flush syringes around the globe.
- For instance, the U.K. Medicines and Healthcare Products Regulatory Agency labels pre-filled syringes for flushing Intravenous (IV) cannulas and IV access devices as ‘borderline’ devices.
- The American Association of Anesthesiology issued guidelines that stated not to refill a syringe once used, even for the same patient.
- In January 2020, the Ministry of Health and Family Welfare of the Government of India provided guidelines for infection prevention and control in healthcare facilities, in which there is a guideline that states never use infusion supplies such as needles, syringes, flush solutions, administration sets, or IV fluids on more than one patient.
Therefore, all these factors fuel the demand for prefilled syringes for flushing across the globe.
RESTRAINING FACTORS
Increasing Recalls of Flush Syringes to Hinder Market Growth
Prefilled syringes have witnessed noteworthy developments in the past decade as preferred container closure systems for flushing the IV catheter. However, the complex designing and manufacturing process, stringent quality assurance, and stability testing methods, are significant challenges faced by the manufacturers.
Thus, owing to such rigorous quality checks, the IV flush manufacturers have recalled multiple products in the past. Since 2006, more than 30 million prefilled IV flush syringes have been recalled. The product recalls were commissioned due to several reasons and some were mild reasons such as mislabeling, improper packaging, while others were severe reasons such as bacterial contamination.
- For instance, in April 2020, BD (Becton, Dickinson, and Company) recalled multiple lots of PosiFlush SF (Sterile Field) saline flush because of holes in the packaging, which may impact and compromise a sterile field. Although the sterility of the outer surface of the syringe may be compromised, the saline solution of the syringe was not impacted. The BD recalled the product distributed between 28 February 2019 through 2 March 2020.
Additionally, recall of prefilled IV flush syringes due to the defect in the working mechanism of syringes or stability concerns of the product is expected to impact the market growth during the forecast period.
- For instance, in August 2021, FDA recalled Cardinal Health’s Monoject Flush Prefilled Syringes containing 0.9% sodium chloride (12mL Syringe with 10mL Saline Fill, 3mL Saline Fill, 5mL Saline Fill). The product recall was due to the possibility of the syringe’s plunger pulling air into the syringe after the health care provider has pushed out (expelled) the air. Injecting air accidentally causes embolism and may result in lethal consequences.
SEGMENTATION
By Type Analysis
To know how our report can help streamline your business, Speak to Analyst
Hardware Segment to Dominate Supported by Advent of New Advanced Products
By type, the global IV flush syringe market share is bifurcated into saline and heparin.
The saline segment dominated the global market share during 2020 on account of the increasing number of inpatient admissions resulting in the need for flush due to multiple medications being administered in chronic conditions, especially chemotherapy (cancer).
- For instance, according to National Cancer Institute, there were approximately 16.9 million cancer patients in the U.S. by 2019, and the patient number is likely to increase to 22.2 million by 2030. Therefore, the demand for flush syringes is anticipated to increase during the forecast period.
Furthermore, the strong product portfolios of market players and the introduction of innovative products in the segment would also contribute to the higher demand.
The heparin segment is anticipated to hold the second-highest share of the market during the forecast period. The segmental growth is mainly due to the usage of heparin flush syringes for patients undergoing angioplasty, heart valve surgery, and others. The use of heparin flush helps prevent blood clots and other complications related to medication administration in these patients. For instance, according to OECD statistics, in 2020, around 13,623 coronary artery bypass surgery was performed in Italy.
By End User Analysis
Increasing Adoption of Flush Syringes in Hospitals & Ambulatory Surgical Centers to Boost Segmental Growth
On the basis of end user, the market is segmented into hospitals & ambulatory surgical centers, clinics, and others.
The hospitals & ambulatory surgical centers segment held the dominating share in 2020. Growing hospital admissions worldwide owing to the increase in the prevalence of chronic diseases is mainly attributable to the higher demand for pre-filled syringes. This factor led to higher segmental growth in 2020.
Additionally, a higher length of hospital stay in the region is majorly responsible for the requirement of the number of flushes per day, thereby increasing the usage frequency of flush syringes. Thus, the factors mentioned above are likely to drive the hospital segment market growth during the forecast period.
REGIONAL INSIGHTS
North America IV Flush Syringe Market Size, 2025 (USD Million)
To get more information on the regional analysis of this market, Download Free sample
On the basis of region, the global market is segmented into North America, Europe, Asia Pacific, and the Rest of the World.
North America
The North America market accounted for USD 161.59 million in 2025, representing 44.78% of the global industry, and is expected to reach USD 171.28 million in 2026. The significant adoption of advanced products in the U.S., increasing inpatient admissions for management of chronic diseases, and the increasing number of surgical procedures in the region are some of the significant factors projected to boost the market’s growth in North America by 2028. For instance, according to the American Lung Association, in 2020, approximately 16.0 million people were suffering from COPD in the U.S. An exacerbation in COPD patients leads to hospital admission demanding special care. This factor is anticipated to contribute to the high demand for these syringes. The U.S. market is projected to reach USD 155.38 million by 2026.
Europe
Europe recorded a market size of USD 113.28 million in 2025, capturing 31.39% of the global market share, and is projected to reach USD 118.83 million in 2026. Europe holds the second-largest share in the global market owing to the rapid development of healthcare infrastructure in European countries, the increasing number of surgical procedures, and technological advancements by market players offering innovative products. The growing number of hospitals in the region are the major factors driving the demand for these syringes in the European market. The UK market is projected to reach USD 30.69 million by 2026 and the Germany market is estimated to reach USD 23.51 million by 2026.
Asia Pacific
In 2025, Asia Pacific represented USD 66.97 million, accounting for 18.56% of the worldwide market, and is projected to grow to USD 71.64 million in 2026. The Asia Pacific market is slated to record the highest CAGR, especially in China, Japan, South Korea, India, and Australia. This factor is because a growing number of medical facilities in Asian countries are adopting advanced products, including prefilled syringes owing to their several associated benefits. Additionally, an increase in the number of procedures in the region is mainly responsible for lengthy hospital stays leading to higher usage of the flush syringe. For instance, according to the OECD statistics in 2019, the average length of hospital stay in Japan was 16 days. The Japan market is projected to reach USD 23.81 million by 2026, the China market is estimated to reach USD 15.93 million by 2026, and the India market is projected to reach USD 11.20 million by 2026.
Rest of the World
The Rest of the World region is anticipated to account for a limited market revenue share during the forecast period. The growing prevalence of chronic diseases, the increasing healthcare expenditure in the region, and the development in healthcare infrastructure in the rest of the world region, specifically in Brazil, Mexico, and GCC, are likely to augment the market's growth in these regions in the forthcoming years. Middle East & Africa contributed 5.27% to the global market in 2025, with a valuation of USD 19.03 million, and is projected to reach USD 19.91 million in 2026.
KEY INDUSTRY PLAYERS
Leading Players Expand Their Manufacturing Capabilities to Strengthen Market Presence
The IV flush syringe market is consolidated with players, such as Becton, Dickinson and Company, B. Braun Melsungen AG, and Cardinal Health, holding the dominant share in 2020. Leading players are eyeing acquisition, collaboration, and new product development strategies to strengthen their positions globally.
- For instance, in October 2021, BD (Becton, Dickinson, and Company) announced that it had reinforced the U.S. government's access to safety injection devices by increasing manufacturing capacity. The new syringe and needle manufacturing lines were developed by the public-private partnership between BD and the Department of Health and Human Services' (HHS) Assistant Secretary for Preparedness and Response (ASPR).
Other notable names in the global market are Medline Industries, LP, NiproMedical Corporation, Medefil Inc., and Polymedicure. These market players are prioritizing new product development and launches to expand their market presence.
LIST OF KEY COMPANIES PROFILED:
- BD (Becton, Dickinson and Company) (U.S.)
- B. Braun Melsungen AG (Germany)
- Cardinal Health (U.S.)
- Nipro Corporation (Japan)
- Medline Industries, LP (U.S.)
- Medefil, Inc. (U.S.)
- Polymedicure (India)
- Aquabiliti (U.S.)
- SteriCare Solutions (U.S.)
KEY INDUSTRY DEVELOPMENTS:
- July 2021 - American weekly news magazine Newsweek recognized Medline Industries, LP by mentioning its two products names in its list ‘Best Infection Prevention Products 2021.’ These two products are ‘Prefilled SwabFlush Syringe with SwabCap’ and ‘ReadyPrep CHG.’
- June 2019 – Medefil, Inc. invested in a manufacturing expansion for increasing the efficiency of its manufacturing facility, owing to this, large production of injectable and flushes is possible than previous manufacturing setup.
REPORT COVERAGE
The global IV flush syringe market research report provides detailed industry analysis. It focuses on key aspects, such as an overview of the advanced product, the rising number of several surgeries, key countries, and pricing analysis. Additionally, it includes an overview of reimbursement scenarios for procedures, key industry developments such as mergers, partnerships, & acquisitions, the impact of COVID-19 on the market, and brand analysis. Besides these, the report offers insights into the IV flush syringe market trends and highlights key industry developments. In addition to the aforementioned factors, it encompasses several factors that have contributed to the growth of the market over recent years. The report also covers regional analysis of different segments.
Request for Customization to gain extensive market insights.
Report Scope & Segmentation
|
ATTRIBUTE |
DETAILS |
|
Study Period |
2021-2034 |
|
Base Year |
2025 |
|
Estimated Year |
2026 |
|
Forecast Period |
2025-2034 |
|
Historical Period |
2021-2024 |
|
Unit |
Value (USD million), Volume (million units) |
|
Segmentation |
By Type
|
|
By End User
|
|
|
By Geography
|
Frequently Asked Questions
The global IV flush syringe market size is projected to grow from USD 381.67 million in 2026 to USD 616.60 million by 2034, exhibiting a CAGR of 6.18% during the forecast period.
In 2025, market value in North America stood at USD 161.59 million.
Registering a CAGR of 6.18%, the market will exhibit steady growth in the forecast period (2026-2034).
The saline IV flush syringe segment is expected to lead this market during the forecast period.
The rise in the prevalence of chronic diseases and increase in inpatient admissions are the major factors driving the markets growth.
Becton, Dickinson and Company, B. Braun Melsungen AG, and Cardinal Health are some of the major players in the global market.
North America dominated the market in terms of share in 2025.
The rise in awareness and favorable guidelines for single-use syringes around the globe are expected to drive the adoption of the products.
Get 20% Free Customization
Expand Regional and Country Coverage, Segments Analysis, Company Profiles, Competitive Benchmarking, and End-user Insights.
Related Reports
-
US +1 833 909 2966 ( Toll Free )
-
Get In Touch With Us