Hepatitis B Vaccine Market Size, Share & Industry Analysis, By Type (Single Antigen and Combination), By Distribution Channel (Hospital & Retail Pharmacies, Government Suppliers, and Others), and Regional Forecast, 2026-2034
KEY MARKET INSIGHTS
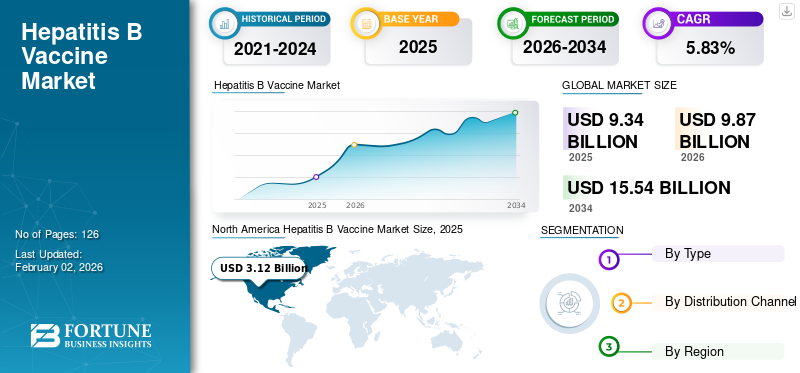
The global hepatitis B vaccine market size was valued at USD 9.34 billion in 2025 and is projected to grow from USD 9.87 billion in 2026 to USD 15.54 billion by 2034, exhibiting a CAGR of 5.83% during the forecast period. North America dominated the hepatitis b vaccine market with a market share of 33.38% in 2025.
The hepatitis B virus (HBV) is a severe public health concern worldwide and a cause of cirrhosis, chronic hepatitis, and hepatocellular carcinoma. There has been a considerable increase in the prevalence of hepatitis B cases due to prenatal transmission from infected mothers to their newborns at birth or in contact with an infected person's body fluids (such as blood or saliva).
- For instance, as per data published by the World Health Organization (WHO), in 2019, around 296 million people suffered from hepatitis B infection. Moreover, 1.5 million new cases are observed annually. (2023)
Thus, owing to the high prevalence of infection, several international public health agencies have implemented strategies to achieve progress toward the global elimination goal of this viral infection.
- For instance, in September 2020, the European Centre for Disease Prevention and Control, with the assistance of a regional action plan implemented by the WHO, dropped the estimated hepatitis B infections to 4.7 million cases in Europe. The regional plan covered 95% of infants with a three-dose vaccine, screening of pregnant women with 90% coverage, and 95% coverage of infants born to infected mothers.
Therefore, the rising prevalence of hepatitis infection and initiatives of mass immunization by government bodies to eliminate the disease led to increasing demand for these vaccines across the globe, further propelling the market growth.
During the COVID-19 pandemic in 2020, the market experienced a decline in its market value due to the lockdown restrictions and a limited number of patients visiting doctors’ clinics. However, the market experienced substantial growth in 2021 and 2022 due to the relaxation of lockdown restrictions and a rise in the number of patients visits to physicians’ offices.
Download Free sample to learn more about this report.
Global Hepatitis B Vaccine Market Overview
Market Size:
- 2025 Value: USD 9.34 billion
- 2026 Value: USD 9.87 billion
- 2034 Forecast Value: USD 15.54 billion, with a CAGR of 5.83% from 2026–2034
Market Share:
- North America led the hepatitis B vaccine market with a 33.38% share in 2025, driven by high disease prevalence and national immunization initiatives.
- By type, the combination vaccine segment held the largest share in 2023 due to better immunogenicity and cost-effectiveness.
- By distribution channel, government suppliers are expected to hold a 65% share in 2025, supported by global vaccination programs and public sector procurement.
Key Country Highlights:
- The hepatitis B vaccine market in Japan is expected to reach USD 374.8 million by 2025.
- China is projected to witness a strong CAGR of 5.80% during the forecast period.
- Europe is anticipated to grow at a CAGR of 6.0%, supported by newborn immunization coverage across 50+ countries.
- The U.S. market continues to expand, aided by FDA fast-track designations and increasing adoption of advanced combination therapies.
Hepatitis B Vaccine Market Trends
High Adoption of Combination Vaccines, especially in Developed Countries
Bacterial or yeast cells are used for manufacturing traditional recombinant vaccines, wherein a piece of DNA is separated from the virus or bacterium and inserted into manufacturing cells.
The use of recombinant DNA technology in vaccines can aid in the production of an optimal immune response for hepatitis B. It can be cost-effective if manufactured using yeast-based recombinant proteins. This also helps in enhancing the safety of the product. However, the traditional single-antigens act only against all known subtypes of the virus with less seroprotection in adults.
- For instance, in February 2023, Dynavax Technologies, a biopharmaceutical company, announced that the U.K.’s Medicines and Healthcare Products Regulatory Agency (MHRA) granted Marketing Authorization for HEPLISAV B, which is a recombinant hepatitis B vaccine for active immunization in adults in the age group of 18 years and older.
Similarly, the production of combination vaccines uses more than one drug to treat a disease. The use of combination therapy has demonstrated more efficacy in patients suffering from hepatitis B than single antigen vaccines.
- In November 2021, Vir Biotechnology, Inc. announced new clinical data from ongoing trials of VIR-2218 in combination with PEG-IFN-α, which demonstrated substantial reductions of HbsAg compared to VIR-2218 alone.
- For instance, in July 2021, Vir Biotechnology, Inc. initiated the phase 2 clinical trials of the VIR-2218 and VIR-3434, a siRNA combination of vaccines to treat chronic viral hepatitis infection.
Also, the approval of the products that have to be administered in two doses contributes to the faster and higher response rate of these vaccines.
- In January 2021, Gilead Sciences, Inc. collaborated with Vir Biotechnology, Inc. to develop a new combination therapy for treating patients with the hepatitis B virus. The main aim of this collaboration is to evaluate different novel therapeutic combinations for developing a new hepatitis B vaccine.
Thus, the rising approval and adoption of combination vaccines compared to traditional ones aid the growth of the global market.
Download Free sample to learn more about this report.
Hepatitis B Vaccine Market Growth Factors
Extensive Investments in R&D by Major Industry Players to Boost Market Growth
The introduction of innovative products in the market is increasing owing to the rising adoption of new drug mechanisms for vaccine production. This will further provide opportunities for companies equipped with strong innovative R&D capabilities to invest heavily in developing new products. Therefore, such investments in R&D by key players for new vaccine trials and production across the globe are further projected to propel the market growth.
- For instance, in December 2022, according to Vir Biotechnology, Inc.’s annual report statistics, the company invested USD 474.6 million in the R&D of new candidates in the pipeline. Currently, the company has four candidates in the pipeline for hepatitis B and all are in phase II of the clinical trials.
Also, the companies are focusing on strategic partnerships to launch a product with high market potential, allowing companies to expand their product portfolio with a strategy to market their product globally. These collaborations will also lead to growth in international sales by registering existing products in new countries and developing new products in highly regulated markets.
- For instance, in June 2021, Vaxelis was launched in the U.S. and was the first hexavalent vaccine approved in the country. Vaxelis is a hexavalent combination vaccine that protects against six diseases, diphtheria, tetanus, pertussis, polio, Hib, and hepatitis B, and was developed as part of a U.S.-based partnership between Merck & Co., Inc., and Sanofi Pasteur.
- Similarly, in September 2022, Valneva SE and VBI Vaccines Inc. announced a partnership in selected European markets for the marketing and distribution of PreHevbri, the only 3-antigen vaccine approved in Europe.
Thus, an increase in investments in the R&D of new vaccines and collaborations of different market players to develop a vaccine with an advanced mechanism of action is anticipated to contribute to the market's growth.
RESTRAINING FACTORS
Stringent Regulatory Policies for Vaccine Approval to Hinder Market Growth
Vaccines under the U.S. Food and Drug Administration (FDA) regulations undergo several rigorous review procedures by laboratories to ensure these products' purity, safety, efficacy, and potency. Also, the approved marketing products have to undergo additional studies for evaluation.
For instance, a manufacturer has to file a Biologics License Application (BLA) after the criteria of phase 3 clinical trials are met, which includes submitting thousands of pages of preclinical and clinical data with details of the manufacturing process of the facility.
Also, manufacturing companies must submit requests to the Stringent Regulatory Authority (SRA) for permission to share nonpublic information with the national regulatory authorities.
- According to the 21 CFR 610.1 regulation of the U.S. FDA, the manufacturer has to test each lot of the product to show confirmation of standards applicable to that product. In order to comply with the standard regulation, the company has to do it on a regular basis for each lot.
Thus, developing a vaccine may take around 10 to 15 years to establish its safety and efficacy. Similarly, the factors contributing to such a long process may further arise with complexities at the time of the development process, the clinical trial process, and different regulatory requirements in other regions. Thus, this process with high time consumption will contribute to additional time utilization owing to stringent regulatory policies by various administrative organizations for approval. Therefore, these factors are expected to limit the introduction of new products by advanced technologies, further restraining market growth.
Hepatitis B Vaccine Market Segmentation Analysis
By Type Analysis
High Potential Advantages of Combination Vaccines to Augment Market Growth
The market is segmented by type into single antigen and combination.
Among types, the combination segment is expected to account for 61.18% of the market in 2026.The dominance was attributed to potential advantages over a single vaccine, such as improved cost-effectiveness, high immunogenicity, and protection against five or more diseases, including hepatitis B. Moreover, these vaccines require administering one or two doses, leading to early disease protection compared to single antigens, further increasing their adoption among the population.
- For instance, in February 2023, VBI Vaccines Inc., a biopharmaceutical company, announced interim data from the Phase 2 study evaluating the effect of combining VBI-2601 (BRII-179), VBI Vaccines Inc.’s HBV immunotherapeutic candidate, and BRII-835 (VIR-2218), an HBV-targeting siRNA candidate, in chronically infected HBV patients.
By type, the single antigen segment is projected to generate USD 3.65 billion in revenue by 2025. Furthermore, rising strategic initiatives by government health bodies across the globe and expanding the role of combination vaccines in population-based vaccination programs with younger age groups further augment the demand for these products.
Thus, the rising demand for these vaccines owing to better potential advantages over single antigens and increasing combination vaccine-based vaccination programs across the globe propel the segment share in the market.
To know how our report can help streamline your business, Speak to Analyst
By Distribution Channel Analysis
Increasing Immunization Rates will Augment the Hospitals & Retail Pharmacies Segment
Based on distribution channel, the market is segmented into hospitals & retail pharmacies, government supplies, and others.
In 2026, the hospitals & retail pharmacies segment is projected to lead the market with a 64.69% share.The highest share was attributed to the large volume of product sales owing to the first vaccination dose administered before the discharge of newborn infants from hospitals. Moreover, the extensive supply of vaccines from hospitals is expected to surge the vaccination uptake amongst the population, further propelling the segment share. By distribution channel, the government suppliers segment is expected to hold a 65% share in 2025.
The government suppliers segment is expected to grow at a significant CAGR during the forecast period. The higher growth was due to rising strategic initiatives by government bodies, such as the World Health Organization (WHO), PAHO, and Ministry of Health and Family Welfare (MoHFW), among others, to eliminate hepatitis B infection across the globe by introducing national and global health programs providing free-of-cost immunization. Moreover, high procurement of vaccines from key players to boost supply and primary vaccination among the population will further propel the segment growth.
- For instance, the Department of Health & Human Services January 2021 announced the Viral Hepatitis National Strategic Plan for the U.S., “A Roadmap to Elimination” (2021–2025), to eliminate viral hepatitis as a public health threat in the country.
REGIONAL INSIGHTS
On the basis of region, the market is studied across North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa.
North America Hepatitis B Vaccine Market Size, 2025 (USD Billion)
To get more information on the regional analysis of this market, Download Free sample
North America
The market in North America reached USD 3.12 billion in 2025, representing 33.38% of total market revenue, and is projected to reach USD 3.27 billion in 2026. The rising prevalence of hepatitis B diseases and the measures taken by national healthcare organizations for prevention, screening, and reducing the mortality rates caused by viral hepatitis infections have been influential for the highest share of the North American region in the global market. Moreover, increasing collaboration among manufacturing players and rising approvals of new combination vaccines with extensive regional presence further boosted the North American market. The US market is projected to reach USD 2.96 billion by 2026.
- In February 2024, GSK plc announced that the U.S. Food and Drug Administration (FDA) had granted Fast Track designation for bepirovirsen, an investigational antisense oligonucleotide (ASO) for the treatment of chronic hepatitis B (CHB).
Europe
Europe contributed approximately USD 2.02 billion to the global market in 2025, accounting for 21.66% share, and is expected to reach USD 2.14 billion in 2026. The market in Europe held a significant share due to a number of regional action plans implemented by European government authorities in order to increase the coverage of newborn neonates against hepatitis B infection. For instance, as per World Health Organization estimates, by December 2019, 50 out of 53 countries in Europe provided routine vaccination with hepatitis B to all infants or children aged 1-12 years, including 23 countries that offered doses to all newborns. Europe is anticipated to grow at a CAGR of 6.0% during the forecast period. The UK market is projected to reach USD 0.42 billion by 2026, while the Germany market is projected to reach USD 0.58 billion by 2026.
Asia Pacific
In 2025, the Asia Pacific market stood at USD 3.05 billion, representing 32.63% of global demand, and is projected to grow to USD 3.24 billion in 2026. The Asia Pacific hepatitis b vaccine market share is driven by the rising prevalence of the disease, with the focus of the vaccine manufacturing players on raising funds and collaborations for the development of novel vaccines, which is anticipated to boost the growth of the region at the highest CAGR over the forecast period. For instance, in April 2022, Shionogi & Co., Ltd., a Japanese research-driven pharmaceutical company, collaborated with NEC Corporation to research and develop a novel vaccine. The collaboration aims to meet the medical needs of the disease condition and to achieve a complete cure for hepatitis B. The hepatitis B vaccine market in Japan is expected to reach USD 374.8 million by 2025. China is projected to witness a strong CAGR of 5.80% during the forecast period. The Japan market is projected to reach USD 0.4 billion by 2026, the China market is projected to reach USD 0.65 billion by 2026, and the India market is projected to reach USD 0.54 billion by 2026.
Rest of the World
Rest of the World maintained a strong presence in the global market, reaching USD 1.15 billion in 2025, accounting for 12.32% share, and is expected to reach USD 1.22 billion in 2026.
On the other side, the Rest of the World (RoW) market, which includes Latin America and the Middle East, is likely to grow at a slower pace. The rising prevalence of hepatitis B infection in the region and the adoption of different strategic frameworks are accelerating the growth of the market in the region.
- For instance, in July 2022, the World Health Organization (WHO) adopted the strategic framework of 2021-2030 in response to the rising prevalence of infection in Africa. The strategy aims to include the birth dose of the hepatitis B vaccine in 35 states of the region.
To know how our report can help streamline your business, Speak to Analyst
List of Key Companies in Hepatitis B Vaccine Market
Robust Investments in New R&D Centers by Key Players to Propel Market
The market is primarily dominated by key manufacturers such as Sanofi and Gilead Sciences, Inc., among others. Owing to the high demand for vaccines worldwide, the top market players are focusing on accelerating investments in vaccine production centers and rising collaborations and acquisitions. These factors are anticipated to augment the growth of the hepatitis B vaccine market.
- In April 2021, Sanofi declared an investment of USD 476.1 million over five years to create a unique vaccine production site in Singapore, pushing the boundaries of operations through cutting-edge manufacturing and digital technologies.
Similarly, other prominent players in the market, including Dynavax Technologies Corporation and Merck & Co., Inc., are continuously engaged in key developments, such as enhancing their distribution channel across the global market through strategic partnerships and extensive product approvals. Such initiatives by key players are projected to propel the global market and register growth by 2029.
- For instance, in November 2021, PreHevbrio was approved by the U.S. Food and Drug Administration (FDA) and is the only approved three-antigen vaccine for adults manufactured by VBI Vaccines Inc. It is commercially available as Sci-B-Vac in Israel and 14 other countries.
LIST OF KEY COMPANIES PROFILED:
- GSK plc (U.K.)
- DYNAVAX TECHNOLOGIES (U.S.)
- VBI Vaccines Inc. (U.S.)
- Sanofi (France)
- Merck & Co., Inc. (U.S.)
- Beijing Minhai Biological Technology Co., Ltd. (China)
- Gilead Sciences, Inc. (U.S.)
- Beijing Tiantan Biological Products Co., Ltd. (China)
- Amy Vaccine Co., Ltd. (China)
- Biokangtai (China)
KEY INDUSTRY DEVELOPMENTS:
- July 2023 - VBI Vaccines Inc. announced the availability of PreHevbri, a hepatitis B vaccine, in the Netherlands and Belgium for active immunization against infection caused by all known subtypes of the hepatitis B virus in adults.
- September 2022 - Valneva SE. and U.S.-based VBI Vaccines Inc. announced a partnership for the marketing and distribution of the PreHevbri vaccine in selected European markets.
- April 2022 - Shionogi & Co., Ltd. and NEC Corporation announced the execution of a strategic research collaboration agreement to develop a hepatitis B therapeutic vaccine.
- December 2021 - Sinovac invested USD 155.0 million in the company's research and development programs, which was comparatively greater than the previous year, accounting for USD 48.8 million in 2020 and USD 24.3 million in 2019. This strategy will assist the company in ensuring its future growth.
- August 2021 - Sanofi announced the acquisition of Translate Bio Inc. to accelerate the adaptability of mRNA technology for the research and development of new vaccines. This strategic acquisition brought advancement to the company's R&D facility and improved the productivity of the products in the pipeline.
REPORT COVERAGE
The research report provides a detailed market analysis and focuses on crucial aspects such as leading players, product types, and major indications of the product. Additionally, it offers insights into market trends and key industry developments such as mergers, partnerships, & acquisitions, and the impact of COVID-19 on the market. In addition to the factors mentioned above, the report includes the factors that have contributed to the market's growth in recent years with a regional analysis of different segments.
Request for Customization to gain extensive market insights.
Report Scope & Segmentation
|
ATTRIBUTE |
DETAILS |
|
Study Period |
2021-2034 |
|
Base Year |
2025 |
|
Estimated Year |
2026 |
|
Forecast Period |
2026-2034 |
|
Historical Period |
2021-2024 |
|
Unit |
Value (USD billion) |
|
Growth Rate |
CAGR of 5.83% from 2026-2034 |
|
Segmentation
|
By Type
|
|
By Distribution Channel
|
|
|
By Region
|
Frequently Asked Questions
Fortune Business Insights says that the global market size was USD 9.34 billion in 2025 and is projected to reach USD 15.54 billion by 2034.
Registering a CAGR of 5.83%, the market will exhibit steady growth over the forecast period (2026-2034).
The combination segment is expected to lead this market during the forecast period.
The rising prevalence of hepatitis B, and the increasing clinical trials for new products across the globe are the key factors driving the market growth.
GSK plc, Sanofi, and Merck & Co., Inc. are the major players in the global market.
High demand for mass immunization and product approvals around the globe are expected to drive the adoption of these products.
Get 20% Free Customization
Expand Regional and Country Coverage, Segments Analysis, Company Profiles, Competitive Benchmarking, and End-user Insights.
Related Reports
-
US +1 833 909 2966 ( Toll Free )
-
Get In Touch With Us