Cancer Vaccines Market Size, Share & Industry Analysis, By Type (Preventive and Therapeutic), By Technology (Molecular-based, Vector-based, and Cell-based), By Indication (Cervical Cancer, Bladder Cancer, Prostate Cancer, Lung Cancer, and Others), By Distribution Channel (Hospitals, Government & Organization Supply, and Others), and Regional Forecast, 2026-2034
KEY MARKET INSIGHTS
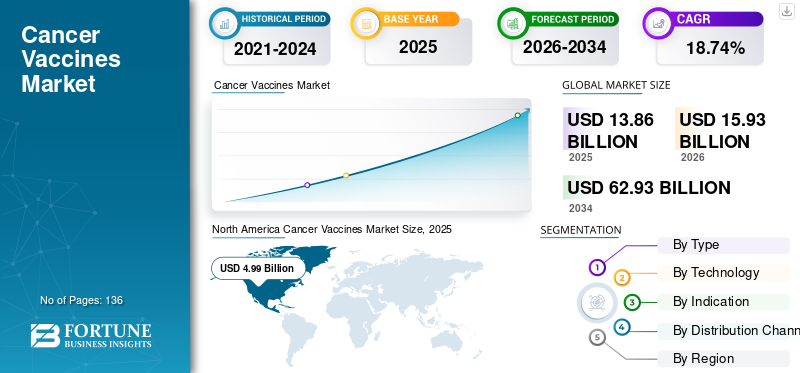
The global cancer vaccines market size was valued at USD 13.86 billion in 2025 and is projected to grow from USD 15.93 billion in 2026 to USD 62.93 billion by 2034, exhibiting a CAGR of 18.74% during the forecast period. North American dominated the cancer vaccines market with a market share of 35.97% in 2025.
Cancer is a significant global public health concern, and with the increasing incidence of cancer globally, cancer prevention has become one of the most critical public health challenges of the 21st century.
- For instance, according to the Cancer Facts & Figures Report published in January 2023 by the American Cancer Society (ACS), an estimated 1.96 new cancer cases in the U.S. would lead to 0.61 million cancer deaths in 2023. The new cancer cases in 2023 indicate an increase of 2.2% compared to 2022.
- Similarly, according to the American Cancer Society, in the U.S., an estimated 13,820 new cases can be recorded of cervical cancer, with 4,360 deaths amongst women in 2024. Furthermore, as per their statistics, cervical cancer is one of the most common causes of cancer death among American women.
Increasing cancer prevalence among the patient population is further resulting in the growing focus on research and development activities to launch novel vaccines in the market. This, along with a growing focus on strategic acquisitions and mergers among major players to expand their presence, is likely to contribute to the global cancer vaccines market share.
- In March 2023, BioNTech SE collaborated with OncoC4, Inc., a clinical-stage biopharmaceutical company dedicated to the discovery and development of novel biologicals for cancer treatment among patients.
Thus, growing research and development activities for innovative therapeutic and preventive cancer vaccines and increasing pipeline candidates, among others, are some of the major factors supporting the growth of the market globally.
The impact of the COVID-19 pandemic resulted in slower growthin 2020. However, despite a decline in the cancer immunization rate among the population amidst the pandemic, increased awareness and high demand for cancer vaccination among individuals contributed to slower growth on the market.
Download Free sample to learn more about this report.
Cancer Vaccines Market Snapshot & Highlights
Market Size & Forecast:
- 2025 Market Size: USD 13.86 billion
- 2026 Market Size: USD 15.93 billion
- 2034 Forecast Market Size: USD 62.93 billion
- CAGR: 18.74% from 2026–2034
Market Share:
- North America dominated the cancer vaccines market with a 35.97% share in 2025, driven by a high prevalence of cancer, established healthcare infrastructure, and strong presence of major industry players.
- By Type, the Preventive segment held the largest share in 2023, owing to increased HPV-related vaccine adoption, public awareness initiatives, and the growing focus of healthcare authorities and manufacturers on cancer prevention.
Key Country Highlights:
- Japan: Demand driven by cervical and gastric cancer immunization efforts, government-led HPV vaccination programs, and increasing public health investments.
- United States: Significant growth supported by ~2.0 million estimated new cancer cases in 2023, expanded pipeline of preventive and therapeutic vaccines, and strong industry presence (e.g., Merck, Moderna).
- China: Growing cancer burden, rising investments in local vaccine development (e.g., Walvax Biotechnology), and supportive national immunization strategies are fueling demand.
- Europe: Advancing through clinical trials and investments in neoantigen and therapeutic cancer vaccines; countries like Germany, France, and the U.K. lead the charge in R&D and regulatory approval efforts.
The cancer vaccines market is evolving rapidly, driven by the growing integration of biotechnology, artificial intelligence (AI), and mRNA vaccine technologies. Companies are leveraging genomic data and machine learning to design highly personalized cancer immunotherapies that target specific tumor antigens. Furthermore, increasing collaborations between academic research centers and biopharma manufacturers are accelerating vaccine innovation, especially for HPV-related cancers and melanoma.
As awareness of early cancer prevention rises, demand for cost-effective cancer vaccines across emerging markets such as India, China, and Brazil is expected to surge over the next decade.
Recent advancements in mRNA-based vaccines, neoantigen discovery, and cell therapy platforms are transforming the development pipeline for cancer vaccines. AI-based modeling tools now allow researchers to identify specific mutations responsible for tumor growth, enabling faster design of personalized vaccine candidates. Additionally, next-generation adjuvants and delivery systems are improving the immune response durability and safety profiles of preventive and therapeutic cancer vaccines.
Cancer Vaccines Market Trends
Rising Investments in R&D Activities and in Preventive or Prophylactic Vaccines for Cancer Prevention
The challenges inherent in developing vaccines for cancer amongst prominent pharmaceutical & biotech players are high. The traditional approach is unreasonable in terms of costs, innovation, and productivity for commercializing these specialized oncology products. Thus, investments in R&D by the major industry players, leading to a rise in the number of new vaccines achieving market approval. However, to achieve the long-term efficiency of cancer immunotherapies, new approaches for R&D investments and advancement in technological innovations by industry players are necessary.
Furthermore, the increasing prevalence of cancer among patients, along with early prevention of cancer using these vaccines, is driving the focus of key players toward the manufacturing of novel vaccines in the market.
- According to the 2023 article published by the Cancer Center, it was reported that the University of Arizona Cancer Center partnered with investigators from 14 university cancer centers and clinics with an aim to conduct a study testing a combination of vaccines for cancer prevention in Lynch Syndrome across the U.S.
- In April 2021, Sanofi announced a USD 476.2 million investment over five years to establish a unique vaccine production center in Singapore, aiming to enhance operations through cutting-edge manufacturing and digital technologies. In partnership with the Singapore Economic Development Board (EDB), the new site will mainly supply the Asian region and complement existing manufacturing capacities in North America and Europe.
Moreover, market players are raising funds from investors and stakeholders to successfully develop a portfolio of vaccine products and anticipate the growth of the market.
- In December 2021, PDC Line Pharma, a clinical-stage biotech company, completed its series B2 round of financing, raising a total of USD 20.3 million with Belgian and Korean investors for cancer vaccine research.
Along with the increasing investments, the number of clinical trials for preventive vaccines has also increased in recent years. For instance, in February 2023, the National Cancer Institute published that the researchers have initiated the clinical trial of preventive vaccines for cancers associated with Lynch Syndrome.
Therefore, rising investments from research institutes and industry players and a boost in vaccine production are anticipated to be critical factors for the global cancer vaccines market growth.
Download Free sample to learn more about this report.
Cancer Vaccines Market Growth Factors
Increasing Incidence of Cancer Among Patients to Support Market Growth
Cancer has emerged as a global public health concern, and with the growing incidence of cancer among patients poses a critical challenges globally.
- According to statistics published by the International Agency for Research on Cancer (IARC) in 2022, it was reported that there were approximately 20 million cancer cases, with lung and breast cancer being the most common among the patient population globally.
- Additionally, according to the same source, it was reported that these cancer cases are projected to increase to 35 million by 2050.
Additionally, certain environmental risk factors, including air pollution and biological risk factors, namely unhealthy diet, alcohol consumption, tobacco use, and smoking, among others, also lead to cancer and other communicable diseases. According to data from the Centers for Disease Control & Prevention (CDC) in 2023, it was reported that almost one-quarter of all cancer in the U.S. deaths are due to lung cancer, with 80% to 90% are linked to cigarette smoking.
- Moreover, in February 2022, the World Health Organization (WHO) stated that cancer-causing infections such as hepatitis and human papillomavirus (HPV), among others, are responsible for approximately 30% of cancer in low and middle-income countries.
However, the increasing strategic initiatives by the government to promote mass immunization are resulting in the adoption of vaccines and further reducing the cancer mortality rate among patients.
Furthermore, about 30-50% of cancers can be prevented by avoiding risk factors and implementing evidence-based prevention strategies such as early screening tests and clinically approved immunization in cancer-developing patients. Therefore, increasing clinical studies among manufacturers on cancer prevention with an aim to launch advanced novel preventive and prophylactic vaccines is likely to support the adoption of these vaccines in the market.
The rising incidence of cancer and high demand for vaccines for the prevention or treatment of cancer across the globe will drive market growth.
Governments across the globe are strengthening national immunization programs and funding large-scale awareness campaigns to combat rising cancer cases. For example, several countries in Europe and Asia Pacific have integrated HPV vaccination into public health policies to reduce cervical cancer incidence. Similarly, organizations such as the World Health Organization (WHO) and Global Alliance for Vaccines and Immunization (GAVI) are focusing on equitable vaccine access, particularly in low- and middle-income countries. These efforts are expected to positively influence the overall cancer prevention ecosystem and improve global survival rates.
RESTRAINING FACTORS
Advances in Personalized Medicine to Limit the Adoption of Preventive and Therapeutic Vaccines
Cancer has become one of the leading threats affecting several million populations globally. Additionally, cancer vaccines, including therapeutic and preventive vaccines, are a type of immunotherapy with limited product approvals, covering only a few cancer types among patients. Preventive vaccines strengthen the body’s immune system in order to treat cancer, while therapeutic vaccines aim to eradicate cancer cells.
However, few cancer types, including breast, colon, and others, contain specific classifications on the basis of their molecular subtypes among patients. Moreover, the expression of certain markers within the same tumor varies for every individual based on the location or stage of cancer. Therefore, the demand for personalized cancer vaccines is increasing for such cancer types to obtain a better prognosis among patients.
Personalized medicine targets specific genes, often with fewer side effects than other types of treatment among patients. Furthermore, personalized medicine does not affect health cells and focuses on targeting cancer cells only among the patient.
- For instance, in October 2020, according to an article published by UC San Diego Health, it was reported that the three-year survival rate among patients who received personalized cancer therapy was 55% compared to 25% in patients who received therapy that was unmatched or had low degrees of matching.
Moreover, the growing benefits of precision medicine, including the use of specific treatment modalities such as targeted drug therapy or immunotherapy depending on the cancer type and severity among patients, is further resulting in the growing number of clinical trials for these vaccines. Along with this, increasing preference toward personalized vaccines owing to advancements in precision medicine, including customized therapy for specific protein and gene changes among patients, is likely to hamper the adoption of these vaccines in the market.
Cancer Vaccines Market Segmentation Analysis
By Type Analysis
Preventive Segment Dominated Owing to Rising Adoption of Vaccines Across the Globe
By type, the global market for cancer vaccines is segmented into preventive and therapeutic.
The preventive segment is anticipated to hold a dominant market share of 91.52% in 2026. The dominance of the segment is due to growing initiatives about the prevention of cancer among manufacturers, government bodies, and various healthcare organizations, resulting in growing demand for preventive vaccines in the market. This, along with a rising focus of key players toward research and development activities to launch novel vaccines is likely to support the growth of the segment in the market globally.
- For instance, in October 2023, Nouscom, a clinical-stage immuno-oncology company, collaborated with the National Cancer Institute with an aim to evaluate positive Phase 1b trial results for the NOUS-209 cancer vaccine at the Society for Immunotherapy of Cancer annual meeting.
On the other hand, the therapeutic segment is expected to reach a comparatively higher CAGR during the forecast period. The growth of the therapeutic segment is attributed to high cancer prevalence globally and rising research initiatives by leading players to curb the rising cancer incidence by introducing new products in the market.
- In June 2022, Treos Bio Limited, a biotechnology company, announced the first patient dosed in a phase 2 trial of polypepi1018 in combination with pd-l1-inhibitor manufactured by F. Hoffmann-La Roche AG for the treatment of metastatic colorectal cancer.
To know how our report can help streamline your business, Speak to Analyst
By Technology Analysis
Molecular-based Segment Dominated the Market Owing to Expansion of Pipeline Candidates
Based on technology, the market is segmented into molecular-based, vector-based, and cell-based.
The molecular-based segment is anticipated to hold a dominant market share of 92.27% in 2026. The highest share was attributable to growing technological advancements in these molecular-based vaccines, such as recombinant vaccines and others, resulting in growing demand for these vaccines among the patient population, thereby supporting the segmental growth in the market. This, along with the growing number of pipeline candidates for molecular-based vaccines, is also likely to contribute to the growth of the segment during the forecast period. This segment is expected to capture 92% of the market share in 2025.
- For instance, in December 2023, Merck & Co., Inc., in collaboration with Moderna, Inc., announced the beginning of the cancer vaccine and KEYTRUDA combo trial in non-small cell lung cancer (NSCLC). With the success of this trial, patients will have the best chance for better outcomes to help fight this disease at earlier stages.
On the other hand, the vector-based segment is also expected to grow during the forecast period. The growth is owing to the growing focus of key players toward launching advanced vector-based vaccines, resulting in the rising adoption of these vaccines among patients, further supporting the segmental growth in the market. This segment is likely to record a significant CAGR of 15.93% during the forecast period (2024-2032).
- In January 2023, Ferring B.V. announced the fully availability of ADSTILADRIN (nadofaragene firadenovec-vncg) across the U.S. for healthcare professionals to prescribe. It is intended for adult patients with high-risk bacillus calmette-guérin (BCG)-unresponsive non-muscle invasive bladder cancer (NMIBC) with carcinoma in situ (CIS) with or without papillary tumors. With significant manufacturing investments, the company achieved a full product supply ahead of schedule.
By Indication Analysis
Cervical Cancer Segment Dominated Owing to Rising Number of Cervical Cancer Cases Amongst Women
By indication, the market is segmented into cervical cancer, bladder cancer, prostate cancer, lung cancer, and others.
The cervical cancer segment is anticipated to hold a dominant market share of 88.89% in 2026. The dominance is due to the growing prevalence of cervical cancer along with rising awareness regarding the condition, resulting in a growing diagnosis rate among women. The growing diagnosis rate is further supporting the rising demand for preventive and therapeutic vaccines for the treatment of cervical cancer globally. The segment is expected to attain 89% of the market share in 2025.
- For instance, according to the World Health Organization (WHO), cervical cancer is the 4th most commonly occurring cancer in women, and there were more than 660,000 new cases recorded in 2022.
Additionally factors, including HPV infections, increased rate of smoking, and prolonged use of oral contraceptives in women in developed countries, is also contributing to the growing number of cervical cancer among patients. Similarly, growing strategic initiatives from governments and companies, aim to raise awareness about the safety and benefits of HPC vaccines, especially in emerging countries, is also likely to support the growth of the segment in the market.
On the other hand, prostate cancer segment is also expected to grow during the forecast period, driven by rising focus of key players’ toward launching novel cancer vaccines for the treatment of prostate cancer and the growing number of pipeline candidates.
Additionally, other segments, including lung cancer and skin cancer, are also expected to grow during the forecast period. The growth is due to research initiatives of clinical trials by leading market players to launch cancer vaccines for skin cancer and liver cancer. This results in the growing adoption of immunotherapeutic approaches toward cancer treatment in the market.
The bladder cancer market is likely to document a substantial CAGR of 22.68% during the forecast period (2024-2032).
By Distribution Channel Analysis
Hospitals Segment Dominated Owing to Increasing Outpatient Visits and Easy Availability of Vaccines in Hospitals
Based on distribution channel, the market is segmented into hospitals, government & organization supply, and others.
The hospitals segment is anticipated to hold a dominant market share of 55.39% in 2026. The dominance was attributed to increasing cancer prevalence and high outpatient visits by individuals in hospitals for primary screening and consultations regarding cancer. Moreover, the high availability and supply of products such as vaccines in hospital settings and patients' preference for vaccine uptake in nearby and well-equipped healthcare centers, further propelled the segment share. This segment is anticipate to gain 55% of the market share in 2025.
- For instance, according to the American Hospital Association’s “2020 Hospital Statistics Report”, in 2018, 879.6 million people visited outpatient departments of hospitals for primary diagnosis and consultations in the U.S.
The government & organization supply segment is expected to grow at a considerable CAGR of 16.36% during the forecast period. The growth is attributed to rising strategic initiatives by government bodies to supply vaccines owing to high cancer prevalence and increasing partnerships with industry players to procure preventive vaccines for mass immunization amongst the population.
- For instance, according to the data published by the United Nations International Children’s Emergency Fund (UNICEF) in 2023, it was reported that they procured 150 million doses of BCG cancer vaccine with the aim of distributing these vaccines among the patient population globally.
Future Outlook
The future of the cancer vaccines market looks promising, with several vaccine candidates entering late-stage clinical trials. Technological convergence combining AI-driven drug discovery, CRISPR gene editing, and nanoparticle-based delivery systems is expected to revolutionize cancer vaccine formulations. Additionally, partnerships between global pharmaceutical companies and regional biotech firms will play a crucial role in expanding access to innovative therapies. By 2032, preventive and therapeutic vaccines are expected to become integral to standard oncology treatment protocols, providing patients with personalized, long-lasting protection.
REGIONAL INSIGHTS
Based on geography, the market is segmented into North America, Europe, Asia Pacific, Latin America, and Middle East & Africa.
North America
North America Cancer Vaccines Market Size, 2025 (USD Billion)
To get more information on the regional analysis of this market, Download Free sample
The market in North America reached USD 4.99 billion in 2025, representing 35.97% of total market revenue, and is projected to reach USD 5.71 billion in 2026. The growth is attributed to the high prevalence of cancer cases and the presence of technologically developed healthcare infrastructure for cancer treatment across North America. The U.S. market is projected to reach USD 5.21 billion by 2026.
- According to 2023 data published by the National Cancer Institute (NCI), it was estimated that approximately 2.0 million are projected to be diagnosed with cancer in the U.S. Moreover, the presence of significant vaccine manufacturers in the U.S., with extensive new product launches, is further boosting the market during 2024-2032.
Moreover, increasing awareness about novel vaccines, along with the presence of significant market players in the U.S., with extensive new prophylactic vaccine launches, fuels the market. Rising strategies by government agencies for preventive immunization across North America, further boost the market during 2024-2032.
- According to the National Immunization Surveys (NIS), in 2020, 56.4% of adolescent females of age group 13-15 years received three doses of the Human Papillomavirus (HPV) vaccines in the U.S.
Europe
Europe contributed approximately USD 4.08 billion to the global market in 2025, accounting for 29.42% share, and is expected to reach USD 4.69 billion in 2026. The U.K. market continues to grow, projected to reach a market value of USD 0.97 billion in 2025. This is attributable to the increasing prevalence of cancer across Europe and rising expenditure on research and development as an initiative to curb cancer prevalence by launching clinical trials in Europe. Germany is set to hold USD 1.13 billion in 2025, while France is anticipated to gain USD 0.20 billion in the same year. The UK market is projected to reach USD 1.12 billion by 2026, and the Germany market is projected to reach USD 1.3 billion by 2026.
- For instance, in September 2021, OSE Immunotherapeutics announced the optimistic final results of its Phase 3 trial of neoepitope-based cancer vaccine Tedopi, called Atalante 1. These results were presented at the European Society for Medical Oncology (ESMO) Virtual Congress, focusing on HLA-A2 positive patients with advanced non-small cell lung cancer (NSCLC).
Asia Pacific
In 2025, the Asia Pacific market stood at USD 4.12 billion, representing 29.71% of global demand, and is projected to grow to USD 4.77 billion in 2026. On the other hand, Asia Pacific is also expected to grow during the forecast period. The Chinese market is predicted to hold USD 1.33 billion in 2025. The growth is due to a rising focus on government and non-governmental organizations to raise awareness about the safety of these cancer vaccines by organizing various immunization programs among patients. This, along with the growing focus of key players on the clinical trials for these cancer vaccines, is further likely to support the growing adoption of these vaccines among patients, thus supporting the growth of the market in the region. India is expected to gain USD 0.59 billion in 2025, while Japan is set to hold USD 0.43 billion in the same year. The Japan market is projected to reach USD 0.49 billion by 2026, the China market is projected to reach USD 1.56 billion by 2026, and the India market is projected to reach USD 0.68 billion by 2026.
Latin America and Middle East & Africa
The Latin America market accounted for USD 0.39 billion in 2025, representing 2.84% of the global industry, and is expected to reach USD 0.45 billion in 2026. Middle East & Africa maintained a strong presence in the global market, reaching USD 0.28 billion in 2025, accounting for 2.05% share, and is expected to reach USD 0.31 billion in 2026. Latin America and the Middle East & Africa are also growing due to the rise of cancer risk factors such as smoking and tobacco use, leading to high cancer prevalence across the countries. Similarly, developing new R&D centers by leading industry players to produce new vaccines will further boost the demand for the vaccine market in these regions. The GCC market is predicted to be worth USD 0.11 billion in 2025.
List of Key Companies in Cancer Vaccines Market
Significant Investments for Increased Vaccine Production to Propel Market Progress
The global market is primarily dominated by a handful of key manufacturers such as Merck & Co., Inc., GlaxoSmithKline plc, and Dendreon Pharmaceuticals LLC., among others. The dominance of these companies is owing to the rising focus of these players on launching clinical trials for cancer vaccine products. This, along with a growing focus on acquisitions and collaborations among the major players to strengthen its global presence, is also supporting the increasing share of these companies in the market.
- For instance, In July 2023, the company and Moderna, Inc., initiated the phase III randomized clinical trial for the evaluation of V940 (mRNA-4157), an investigational personalized mRNA cancer vaccine, in combination with KEYTRUDA patients with resected high-risk melanoma.
- In March 2024, Merck & Co., Inc., announced its plan to conduct clinical trials of a new investigational multi-valent HPV vaccine intended to provide broader protection against multiple HPV types and single dose regimen for GARDASIL 9 to evaluate the safety and efficacy compared to the approved three-dose regimen.
Similarly, other prominent players in the market, including Walvax Biotechnology Co., Ltd., and Synthaverse S.A., are continuously engaged in the production of these vaccines and are focusing on enhancing their distribution channels globally. Through strategic partnerships and R&D investments, they aim to establish their footprints in emerging nations. Such initiatives are projected to propel the number of emerging players and are expected to register growth by 2032.
LIST OF KEY COMPANIES PROFILED:
- Dendreon Pharmaceuticals LLC. (U.S.)
- Serum Institute of India Pvt. Ltd. (India)
- Merck & Co., Inc. (U.S.)
- GlaxoSmithKline plc (U.K.)
- Walvax Biotechnology Co., Ltd. (China)
- Synthaverse S.A. (Poland)
- Center of Molecular Immunology (Cuba)
- Ferring B.V. (Switzerland)
- Wantai BioPharm (China)
- Dynavax Technologies Corporation (U.S.)
KEY INDUSTRY DEVELOPMENTS:
- March 2024 – BostonGene, NEC, and Transgene expanded their collaboration for the Phase I/II Clinical Trial of Neoantigen Cancer Vaccine TG4050.
- February 2024 – Moderna announced the initiation of a cancer vaccine trial in the U.K. The first U.K. patients received an experimental mRNA vaccine at Imperial College Healthcare NHS Trust.
- November 2021 - Dendreon Pharmaceuticals LLC., a manufacturer of dendritic cell (D.C.) cancer vaccine, and Shoreline Biosciences announced CMC and a manufacturing alliance to advance the future of iPSC cellular therapy.
- December 2020 - Merck & Co., Inc. declared to build a new manufacturing facility to increase its production of TICE Bacillus Calmette–Guérin, tripling its current manufacturing capacity.
- August 2020 - GlaxoSmithKline Korea faced indicted under Korea’s Monopoly Regulation and Fair Trade laws in relation to government tenders of HPV (Cervarix) and PCV (Synflorix) vaccines in 2018 and 2019.
- June 2020 - Vaccine manufacturers Xiamen Innovax Biotech Co., Ltd. and Walvax Biotechnology Co., Ltd. committed to provide enough supply to immunize at least 84.0 million girls in Gavi countries, ahead of Global Vaccine Summit 2020.
Expert Insights
Experts believe that the next generation of cancer vaccines will not only target existing tumors but also help prevent recurrence by boosting the body’s adaptive immune memory. The increasing use of AI-powered biomarker identification and real-world clinical data is streamlining vaccine design and improving efficacy outcomes. As the industry continues to evolve, leading players are expected to focus on scalable manufacturing, global accessibility, and regulatory harmonization to ensure equitable cancer prevention worldwide.
REPORT COVERAGE
The global cancer vaccines market report provides a detailed market analysis and focuses on crucial aspects such as leading players, product types, and major product indications. Additionally, it offers insights into market trends and key industry developments such as mergers, partnerships, & acquisitions. In addition to the factors mentioned above, the report includes the factors that have contributed to the market's growth in recent years with a regional analysis of different segments.
Request for Customization to gain extensive market insights.
Report Scope & Segmentation
|
ATTRIBUTE |
DETAILS |
|
Study Period |
2021-2034 |
|
Base Year |
2025 |
|
Estimated Year |
2026 |
|
Forecast Period |
2026-2034 |
|
Historical Period |
2021-2024 |
|
Growth Rate |
CAGR of 18.74% from 2026-2034 |
|
Unit |
Value (USD billion) |
|
Segmentation
|
By Type
By Technology
By Indication
By Distribution Channel
By Region
|
Frequently Asked Questions
Fortune Business Insights says that the global market size was USD 13.86 billion in 2025 and is projected to reach USD 62.93 billion by 2034.
Registering a CAGR of 18.74%, the market will exhibit steady growth in the forecast period.
The preventive segment dominated the market in 2025.
The rising prevalence of cancer cases and increasing clinical trials for new cancer vaccines across the globe are the key factors driving the market growth.
Key companies include Merck & Co., Inc., GlaxoSmithKline plc, Moderna, Inc., Dendreon Pharmaceuticals LLC, and Walvax Biotechnology Co., Ltd.
High demand for mass immunization and product approvals around the globe is expected to drive the adoption of these products.
Cancer vaccines are biological preparations that stimulate the immune system to recognize and destroy cancer cells, either preventive vaccines or therapeutic vaccines.
Preventive vaccines, particularly HPV and Hepatitis B vaccines, are widely used to prevent cervical and liver cancers, respectively.
Rising cancer incidence, advancements in mRNA technology, government immunization programs, and increasing investment in biotech R&D are key growth factors.
Get 20% Free Customization
Expand Regional and Country Coverage, Segments Analysis, Company Profiles, Competitive Benchmarking, and End-user Insights.
Related Reports
-
US +1 833 909 2966 ( Toll Free )
-
Get In Touch With Us