Endotoxin Testing Reagents Market Size, Share & Industry Analysis, By Product Type (LAL/TAL Reagents, Recombinant Endotoxin Detection Reagents, Standards & Controls, & Others), By Source (Natural, Recombinant, & Others), By Test Method (Gel-clot, Chromogenic, Turbidimetric, Recombinant Methods, & Others) By Application (Pharmaceutical Product Testing, Biologics & Vaccine Testing, Medical Device Testing, Raw Material Testing, Water & Process Sample Testing), By End User (Pharmaceutical & Biotechnology Companies, Medical Device Manufacturers, CROs & CDMOs), & Regional Forecast, 2026-2034
Endotoxin Testing Reagents Market Size and Future Outlook
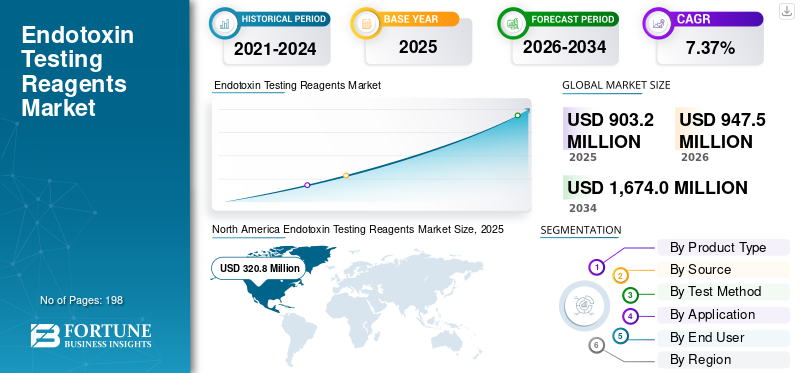
The global endotoxin testing reagents market size was valued at USD 903.2 million in 2025. The market is projected to grow from USD 947.5 million in 2026 to USD 1,674.0 million by 2034, exhibiting a CAGR of 7.37% during the forecast period. North America dominated the endotoxin testing reagents market with a market share of 57.66% in 2025.
Reagents for endotoxin testing are used in quality control processes to identify and measure bacterial endotoxins in various samples. The market growth is propelled by the ongoing expansion of biologics and sterile injectable production, the increasing significance of contamination control in regulated manufacturing, and the escalating move from traditional lysate-based techniques to recombinant endotoxin detection agents. The global market demand is also rising as manufacturers enhance quality assurance measures through batch release testing, in-process monitoring, and raw material qualification to boost product safety, compliance, and manufacturing dependability.
Major players in the market comprise Charles River Laboratories, Lonza, Associates of Cape Cod, and bioMérieux. These firms are focusing on assay sensitivity, regulatory standards, workflow efficiency, and the capability to support quality control environments for pharmaceuticals, biologics, vaccines, and medical devices.
Download Free sample to learn more about this report.
ENDOTOXIN TESTING REAGENTS MARKET TRENDS
Rise of Animal-Free Alternatives is a Significant Trend Observed in the Global Market
The emergence of animal free alternatives is a significant trend in the market, as pharmaceutical and medical device manufacturers increasingly seek ways to reduce their reliance on lysate derived from horseshoe crabs while ensuring test sensitivity, consistency, and regulatory compliance. This transition is facilitating quicker use of recombinant endotoxin detection reagents, particularly in high-value biologics, vaccine, and sterile production processes. Methods that do not involve animals are also becoming popular as they assist companies in enhancing sustainability objectives, increasing supply stability, and minimizing variability associated with natural-source reagents. Consequently, recombinant assays are transitioning from a developing alternative to a more commercially significant technology sector within worldwide QC laboratories. These factors are supporting the overall global endotoxin testing reagents market growth.
- For instance, in January 2024, Charles River Laboratories launched Endosafe Trillium rCR cartridges as the first rapid animal-free bacterial endotoxin test, combining its cartridge platform with recombinant cascade reagent technology to provide a non-animal-derived alternative for endotoxin testing.
MARKET DYNAMICS
MARKET DRIVERS
Download Free sample to learn more about this report.
Continued Expansion of Biologics and Sterile Injectable Manufacturing is Driving Market Growth
Continued expansion of biologics and sterile injectable manufacturing is a major market driver, as each increase in production capacity creates greater demand for batch-release testing, in-process quality control, raw material screening, and water/process sample monitoring. Biologics, vaccines, and sterile injectables require strict contamination control, so as manufacturers add new lines and expand facilities, reagent consumption rises along with testing volumes. This is especially important in regulated manufacturing environments where endotoxin testing is a routine safety requirement before products can move forward in production or release. The driver is becoming stronger as companies invest in larger biologics networks, more advanced sterile manufacturing sites, and higher-value injectable therapies. As a result, the market benefits from higher production output and more frequent, complex QC workflows across global manufacturing sites. All these factors cumulatively drive the overall market growth.
- For instance, in November 2025, AstraZeneca announced it would invest USD 2 billion to expand its Maryland manufacturing footprint, including a significant expansion of its biologics manufacturing facility in Frederick and a new facility in Gaithersburg for the clinical supply of innovative molecules.
MARKET RESTRAINTS
High Cost of Advanced Reagents to Limit Market Growth
The high cost of advanced reagents is a significant market limitation, as recombinant endotoxin detection reagents and rapid cartridge-based systems frequently cost more than numerous traditional lysate-based techniques. This complicates adoption for small and medium-sized manufacturers, contract laboratories, and budget-conscious users, particularly in developing markets where testing finances are more constrained. The increased expense is attributed to the assay kit, validation tasks, modifications in workflow, and sometimes the need for specialized instruments. Consequently, numerous laboratories continue to use traditional LAL/TAL methods despite their interest in adopting newer, animal-free or quicker technologies. This hinders the speed of transition to premium products and restricts revenue growth in segments sensitive to price.
- For instance, on March 18, 2026, Fisher Scientific listed Charles River Endosafe Compendial LAL Cartridges at USD 531.60 for 10 tests (about USD 53.16 per test) and Charles River’s endpoint chromogenic kit at USD 937.62 for 140 tests. These prices highlight that advanced and rapid endotoxin testing solutions can carry a significant cost burden.
MARKET OPPORTUNITIES
Growing Shift from Conventional Lysate-Based Methods Toward Recombinant Reagents to Offer Market Growth Opportunities
The transition from traditional lysate-based techniques to recombinant reagents is generating significant market potential, as numerous pharmaceutical and biotech firms seek endotoxin testing solutions that provide improved batch consistency, enhanced supply reliability, and reduced reliance on materials sourced from horseshoe crabs. This transition is creating new growth opportunities for suppliers capable of delivering validated recombinant assays, automated systems, and workflow-compatible solutions for regular QC applications. It also enables premium pricing opportunities; as recombinant techniques are increasingly viewed as more valuable options instead of mere substitutes. With increased regulatory and industry acceptance of recombinant testing, vendors have greater a chance of transitioning legacy LAL/TAL users in biologics, vaccines, sterile injectables, and medical devices. This trend is significant in extensive manufacturing settings, where assay dependability, scalability, and sustainability are increasingly pivotal purchasing considerations. All these factors would drive the market growth over the coming years.
- For instance, in September 2024, Lonza announced that it had started expanding its endotoxin assay production facility in Walkersville, Maryland. The company stated that the expansion is intended to meet growing demand for endotoxin assay products, which supports the view that there is rising interest in advanced endotoxin testing methods.
MARKET CHALLENGES
Ethical Concerns Regarding the Use of Horseshoe Crab Blood in LAL Testing Pose a Prominent Challenge to Market Growth
Ethical issues surrounding the use of horseshoe crab blood in LAL testing pose a significant market challenge by heightening pressure on manufacturers to simultaneously address patient safety, regulatory compliance, and sustainability demands. Conventional LAL testing relies on lysate derived from horseshoe crab blood, raising concerns about ongoing dependence on this resource for animal welfare and conservation. This generates uncertainty for suppliers who are still significantly reliant on natural-source reagents, particularly as an increasing number of purchasers and regulators embrace animal-free alternatives. It also impacts long-term supply strategy, product placement, and investment focus throughout the market. Consequently, businesses face a challenging shift as traditional techniques remain prevalent while consumer preferences are slowly shifting toward innovative solutions. bioMérieux highlights that the pharmaceutical sector’s dependence on LAL raises ethical dilemmas, as it comes from wild horseshoe crabs, making the issue increasingly crucial for the industry. All the factors cumulatively affect the market growth.
Segmentation Analysis
By Product Type
LAL/TAL Reagents Segment Dominates Due to its Strong Legacy Validation and Broad Routine Use
In terms of product type, the market is divided into LAL/TAL reagents, recombinant endotoxin detection reagents, standards & controls, and others.
The LAL/TAL reagents segment held the largest endotoxin testing reagents market share in 2025. This is driven by its long-standing use in regulated endotoxin testing and its broad acceptance across various sample testing. These reagents continue to hold the largest share as many quality control laboratories already have validated workflows, trained personnel, and established SOPs built around conventional lysate-based methods. Their wide commercial availability, familiarity with routine batch release testing, and continued use in cost-sensitive and legacy manufacturing environments further support segment dominance. Moreover, many manufacturers still prefer LAL/TAL-based assays, as changing to newer methods often requires additional validation work, process adjustments, and regulatory comfort, which slows rapid adoption and supports the segment’s leading position.
- For instance, in February 2024, FUJIFILM Irvine Scientific announced that it had added key LAL products from FUJIFILM Wako Chemicals U.S.A. to its life sciences portfolio, expanding complete endotoxin-testing solutions available directly to customers in the U.S. and Europe.
The recombinant endotoxin detection reagents segment is anticipated to grow with a CAGR of 12.66% over the forecast period.
To know how our report can help streamline your business, Speak to Analyst
By Source
Strong Legacy Acceptance and Broad Installed Base Supported the Dominance of Natural Segment
Based on source, the market is classified into recombinant, natural, and others.
The natural segment captured the leading position in the global market in 2025. Key factors, including its long-established use in bacterial endotoxin testing and its continued acceptance across pharmaceutical products, biologics, vaccines, medical devices, and water/process testing, are supporting the dominance of the segment. Moreover, many manufacturers still prefer natural-source methods, as shifting to recombinant alternatives often requires revalidation, internal process changes, and additional regulatory approvals, which slow conversion and help natural reagents maintain higher sales. Furthermore, the segment is set to hold a 69.2% share in 2026.
- For instance, in 2025, Associates of Cape Cod noted in its endotoxin testing supplement that the industry still uses two primary methods: traditional LAL reagents and recombinant reagents, highlighting the continued central role of traditional natural-source LAL products in current testing practice.
The recombinant segment is anticipated to rise with a CAGR of 12.65% over the forecast period.
By Test Method
Chromogenic Segment Dominated Due to its Fast Quantification and Strong Routine QC Use
Based on test method, the market is classified into gel clot, chromogenic, turbidimetric, recombinant methods, and others.
The chromogenic segment dominated with the largest market share in 2025. This is due to its significant sensitivity, quantitative results, and widespread use in pharmaceuticals, biologics, vaccines, and the testing of medical devices. This approach maintains the highest percentage by enabling laboratories to accurately assess endotoxin levels, thereby facilitating regular quality control and testing for batch release. Its quicker turnaround, greater compatibility with high-throughput workflows, and robust alignment with regulated manufacturing settings further reinforce segment leadership. Additionally, numerous end users favor chromogenic assays due to their effective combination of precision, workflow efficiency, and recognized application in endotoxin testing programs, maintaining strong demand in QC labs worldwide. Furthermore, the segment is set to hold 35.1% share in 2026.
- For instance, Charles River laboratories is one of the leading players in the market that offers products for chromogenic testing.
The recombinant methods segment is anticipated to rise with a CAGR of 12.41% over the forecast period.
By Application
Strong Routine Release and Quality Control Needs Led to Dominance of Pharmaceutical Product Testing Segment
On the basis of application, the market is divided into pharmaceutical product testing, biologics and vaccine testing, medical device testing, raw material testing, water & process sample testing, and others.
In 2025, the pharmaceutical product testing segment dominated the market. This is due to the high volume of sterile and injectable medications that require regular endotoxin testing before distribution. This segment maintains its largest share, as pharmaceutical manufacturers conduct endotoxin testing on finished product batches as a routine part of quality control and compliance processes. The extensive network of pharmaceutical manufacturing facilities, ongoing batch-release obligations, and heightened emphasis on product safety further bolster the segment’s dominance. Additionally, testing pharmaceutical products creates a consistent demand for reagents, as it is a standard, frequently used process in regulated manufacturing settings, thereby maintaining elevated testing volumes worldwide. Furthermore, the segment is set to hold 34.4% share in 2026.
- For instance, in September 2025, Medistri announced the expansion of its services for pharmaceutical vials and stated that its GMP-accredited laboratory would perform Bacterial Endotoxin Testing to demonstrate the safety of pharmaceutical vials.
The biologics and vaccine testing segment is anticipated to rise with a CAGR of 9.39% over the forecast period.
By End User
Pharmaceutical & Biotechnology Companies Led Demand due to Broad Quality Control Use Across Manufacturing
Based on end user, the market is segmented into pharmaceutical & biotechnology companies, medical device manufacturers, CROs & CDMOs, and others.
The pharmaceutical & biotechnology companies segment dominated the market in 2025. This dominance is driven by significant utilization of endotoxin testing reagents for finished drug products, biologics, vaccines, raw materials, and water/process sample analysis. This segment maintains the largest share, as these companies manage the most regulated manufacturing and batch release processes, leading to consistent, ongoing demand for testing reagents. Their intense emphasis on product safety, GMP adherence, and contamination prevention further reinforces their leadership in the market segment. Additionally, the growing adoption of sterile injectables, biologics, and advanced therapies is increasing endotoxin testing requirements within internal quality control laboratories, making pharmaceutical and biotechnology firms the primary end-user segment. Furthermore, the segment is set to hold 65.4% share in 2026.
- For instance, in November 2024, LGM Pharma announced the expansion of its analytical testing services to include endotoxin and rapid sterility testing, stating that these services are essential for pharmaceutical companies developing sterile and injectable products.
In addition, CROs/CDMOs are projected to grow at a 7.11% rate during the forecast period.
Endotoxin Testing Reagents Market Regional Outlook
By geography, the market is divided into Latin America, Asia Pacific, Europe, North America, and the Middle East & Africa.
North America
North America Endotoxin Testing Reagents Market Size, 2025 (USD Million)
To get more information on the regional analysis of this market, Download Free sample
The North American market size reached USD 306.1 million in 2024 and dominated the global market. In 2025, the area maintained its leading share, with USD 320.8 million. Growth in North America is mainly supported by the region’s strong biologics and sterile injectable manufacturing base, high adoption of advanced QC methods, and early shift toward recombinant endotoxin testing reagents. The region also benefits from large pharma and biotech investments in new manufacturing capacity.
U.S. Endotoxin Testing Reagents Market
In North America, the U.S. led the market and is projected to reach approximately USD 290.8 million in 2026, representing about 30.7% of the global market.
Europe
Europe’s market size is anticipated to grow at a CAGR of 6.36% during the forecast period. Europe is driven by its large biopharmaceutical and vaccine manufacturing footprint, strong regulatory quality culture, and rising investment in regional bioproduction capacity. The market also benefits from the modernization of endotoxin testing practices and the broader interest in animal-free methods.
U.K. Endotoxin Testing Reagents Market
The U.K. market in 2026 is estimated at around USD 32.4 million, representing roughly 3.4% of global revenues.
Germany Endotoxin Testing Reagents Market
Germany’s market size is projected to reach approximately USD 42.6 million in 2026, equivalent to around 4.5% of global sales.
Asia Pacific
The Asia Pacific market size is expected to reach USD 243.0 million by 2026. The region is expected to grow at the fastest rate due to rapid expansion in biologics, vaccines, biosimilars, and CDMO manufacturing, along with improvements in QC infrastructure across China, India, Japan, South Korea, and Southeast Asia. The region also benefits from rising export-oriented pharmaceutical production, which increases the need for compliant endotoxin testing.
Japan Endotoxin Testing Reagents Market
The Japanese market in 2026 is estimated at around USD 49.8 million, accounting for roughly 5.3% of global revenues.
China Endotoxin Testing Reagents Market
China’s market is projected to reach revenues of around USD 72.6 million in 2026, representing roughly 7.7% of global sales.
India Endotoxin Testing Reagents Market
The Indian market in 2026 is estimated at around USD 34.5 million, accounting for roughly 3.6% of global revenues.
Latin America and the Middle East & Africa
The growth in the Latin America and the Middle East & Africa regions is anticipated to be slower over the forecast period. The market is driven by increased manufacturing capacity for vaccines and pharmaceuticals, enhanced quality-control systems, and greater involvement in regional and global biologics supply chains. The Latin American market in 2026 is estimated at around USD 68.5 million.
In the Middle East & Africa region, the GCC market is projected to reach approximately USD 23.9 million by 2026, representing about 2.5% of global revenues.
COMPETITIVE LANDSCAPE
Key Industry Players
Major Players Focus on Expanding Reagent Portfolios and Advanced Endotoxin Testing Solutions to Strengthen Market Position
The global market for endotoxin testing reagents is semi-concentrated, with key companies such as Charles River Laboratories, Lonza, Associates of Cape Cod, and bioMérieux representing a significant portion of market revenue. These firms aim to enhance recombinant endotoxin detection options, bolster traditional LAL/TAL reagent collections, and boost assay sensitivity, reliability, and workflow efficiency to reinforce their market position.
- For instance, in June 2025, Charles River partnered with TerraCycle on an Endosafe cartridge recycling program. The program lets users of Endosafe BET cartridge technology recycle single-use cartridges used for routine water testing.
Other significant participants include FUJIFILM Corporation, Merck KGaA, Thermo Fisher Scientific, and regional suppliers such as Bioendo. These companies are expected to prioritize new product innovation, portfolio expansion, and stronger support for regulated quality control environments to improve their competitive positions over the forecast period.
LIST OF KEY ENDOTOXIN TESTING REAGENTS COMPANIES PROFILED
- Charles River Laboratories (U.S.)
- Lonza (Switzerland)
- BIOMÉRIEUX (France)
- FUJIFILM Corporation (Japan)
- Merck KGaA (Germany)
- Associates of Cape Cod, Inc. (U.S.)
- Thermo Fisher Scientific Inc. (U.S.)
- InvivoGen (U.S.)
- Xiamen Bioendo Technology Co., Ltd. (China)
- ACROBiosystems (U.S.)
KEY INDUSTRY DEVELOPMENTS
- May 2025: ACC issued a formal position statement on USP Chapter <86> and recombinant BET reagents. The company said USP <86> marks a major milestone for recombinant endotoxin testing and reiterated that PyroSmart NextGen was the first commercially available rCR reagent.
- August 2024: Associates of Cape Cod (ACC) stated that USP <86> approval positioned it to support broader recombinant BET adoption.
- June 2024: FUJIFILM Wako launched two new pyrogen/endotoxin tests: LumiMAT and PYROSTAR Neo+. PYROSTAR Neo+ is a recombinant reagent for bacterial endotoxin detection.
- May 2024: Bioendo showcased its Phoenix Intelligent Automatic Endotoxin Detection System at CPHI Shanghai 2024.
- March 2022: Bioendo highlighted its new micro kinetic chromogenic endotoxin test kit. It is a dedicated endotoxin reagent kit that offers higher sensitivity, a broader linear range, and compatibility with vaccines, antibodies, and biologics.
REPORT COVERAGE
The global endotoxin testing reagents market analysis includes a thorough evaluation of the market size and forecasts for every segment highlighted in the report. It offers insights into the market dynamics and trends expected to drive the market over the forecast period. It provides understanding of essential factors, including technological progress, product innovations, the regulatory environment, and the launch of new products. Additionally, it details partnerships, mergers, and acquisitions, as well as key developments in the industry within the market. The global market forecast report also provides an in-depth competitive landscape, including information on market share and profiles of key active players.
Request for Customization to gain extensive market insights.
Report Scope & Segmentation
| ATTRIBUTE | DETAILS |
| Study Period | 2021-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2021-2024 |
| Growth Rate | CAGR of 7.37% from 2026-2034 |
| Unit | Value (USD Million) |
| Segmentation | By Product Type, Source, Test Method, Application, End User, and Region |
| By Product Type |
|
| By Source |
|
| By Test Method |
|
| By Application |
|
| By End User |
|
| By Region |
|
Frequently Asked Questions
According to Fortune Business Insights, the global market value stood at USD 903.2 million in 2025 and is projected to reach USD 1,674.0 million by 2034.
In 2025, the North American market value stood at USD 320.8 million.
The market is expected to exhibit a CAGR of 7.37% during the forecast period.
By product type, the LAL/TAL reagents led the market in 2025.
Growing demand for biologics and stringent regulatory requirements for drug safety are the key factors driving the market.
Charles River Laboratories, Lonza, Associates of Cape Cod, and bioMérieux are some of the prominent players in the global market.
North America dominated the market in 2025 with the largest share.
Get 20% Free Customization
Expand Regional and Country Coverage, Segments Analysis, Company Profiles, Competitive Benchmarking, and End-user Insights.
Related Reports
-
US +1 833 909 2966 ( Toll Free )
-
Get In Touch With Us