Immunodiagnostics Market Size, Share and Industry Analysis By Product Instruments, Reagents & Consumables), By Application (Oncology & Endocrinology, Hepatitis & Retrovirus, Cardiac Markers, Infectious Diseases), By End user (Clinical Laboratories, Hospitals, Physician’s Offices), By End-user(Hospitals, Dental Clinics, Academic & Research Institutes) and Regional Forecast, 2026-2034
KEY MARKET INSIGHTS
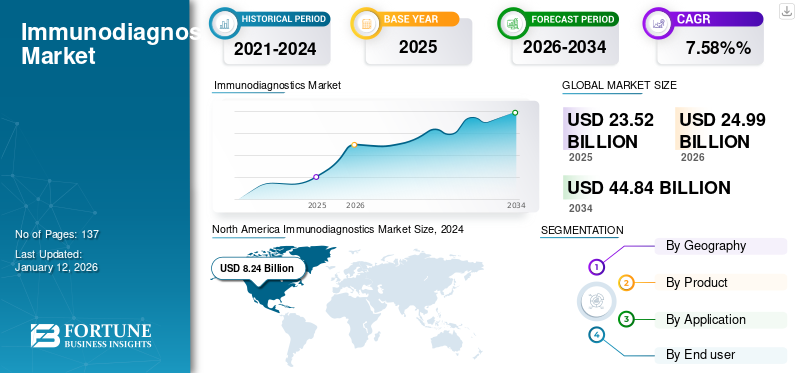
The immunodiagnostics market size was valued at USD 23.52 billion in 2025. The market is projected to grow from USD 24.99 billion in 2026 to USD 44.84 billion by 2034, exhibiting a CAGR of 7.58% during 2026-2034. North America dominated the immunodiagnostics market with a market share of 37.02% in 2025.
Immunodiagnostics is a branch of in-vitro diagnostics with high sensitivity, which has led to the emergence of prospective diagnostic technologies, diagnostic platforms, and new immunoassay formats. The Enzyme-Linked Immunosorbent Assay (ELISA) and Radioimmunoassay (RIA) have been the predominant techniques since the initial era of immunodiagnostics.
The market is expected to grow rapidly during the forecast period, owing to the increasing incidence & prevalence of infectious diseases such as cancer, respiratory infections, hepatitis, HIV infection, and other infectious diseases. The rise in geriatric population, technological advancements in the diagnostic kits and rising utilization of immunodiagnostics in rapid point of care testing devices are some of the factors expected to drive the market during 2025-2032.
Download Free sample to learn more about this report.
"Oncology & Endocrinology is the Attractive Application Type"
Oncology & Endocrinology segment accounted for a market share of 25.6% in 2024. The highest share of the segment is attributed to the adoption of immunodiagnostics in several applications such as cancer and endocrine disease. Increase in the number of new product approvals, and adequate reimbursement for products and tests are expected to drive the overall immunodiagnostics market during the forecast period. For instance, in Nov 2024, OPKO Health Inc. received approval from the U.S. Food and Drug Administration (FDA) for the total prostate-specific antigen test with the immunoassay analyzer.
To know how our report can help streamline your business, Speak to Analyst
In terms of product types, the reagents & consumables segment is likely to be the fastest-growing segment during the forecast period, owing to the high replacement rate and several consumables are single-use products. Based on end users, clinical laboratories segment is likely to be the fastest-growing segment for the forecast duration owing to the gradual shift of testing from hospitals and clinical laboratories.
Download Free sample to learn more about this report.
Global Immunodiagnostics Market Snapshot & Highlights
Market Size & Forecast:
- 2025 Market Size: USD 23.52 billion
- 2026 Market Size: USD 24.99 billion
- 2034 Forecast Market Size: USD 44.84 billion
- CAGR: 7.58% from 2026 to 2034
Market Share:
- Region: North America dominated the market with a 37.02% share in 2025. This leadership is driven by the rising prevalence of infectious diseases, an increasing number of new product approvals, and the presence of adequate reimbursement for products and tests in the region.
- By Application: The oncology & endocrinology segment held the largest market share, accounting for 25.6% in 2024. The segment's dominance is due to the high adoption of immunodiagnostics for cancer and endocrine disease detection, an increase in new product approvals, and adequate reimbursement for these tests.
Key Country Highlights:
- Japan: As a key country in the fast-growing Asia Pacific market, growth is propelled by the higher potential for intense penetration of advanced immunodiagnostic products and a rising emphasis on early disease detection.
- United States: Market growth is fueled by a rising prevalence of infectious and chronic diseases, a high number of new product approvals from the U.S. FDA, and a favorable reimbursement landscape that encourages the adoption of advanced diagnostic tests.
- China: The market is driven by the increasing penetration of immunodiagnostic technologies as the country's healthcare infrastructure develops and the demand for effective and early disease diagnosis grows within the Asia Pacific region.
- Europe: The market is advanced by a strong and growing emphasis on early diagnosis and disease prevention by government organizations. The presence of a well-established healthcare system also supports the high adoption of immunodiagnostic tests.
Regional Analysis
North America Immunodiagnostics Market Size, 2025 (USD Billion)
To get more information on the regional analysis of this market, Download Free sample
"North America Emerges as Lucrative Market for Immunodiagnostics"
North America
North America contributed approximately USD 8.71 billion to the global market in 2025, accounting for 37.02% share, and is expected to reach USD 9.23 billion in 2026. and is anticipated to register a comparatively higher CAGR during 2025 to 2032 due to the rising prevalence of infectious diseases, an increase in the number of new product approvals, and adequate reimbursement for products and tests in the region. Moreover, the aging population, rising healthcare expenditures, increasing emphasis on better patient outcomes, and huge patient pool are the factors considered to support the immunodiagnostics market growth in North America during the forecast period.
At present, developed nations hold a dominant share and are considered to grow slowly compared to emerging markets during the forecast period.
Europe
In 2025, the Europe market stood at USD 5.77 billion, representing 24.52% of global demand, and is projected to grow to USD 6.12 billion in 2026.
Asia Pacific
The Asia Pacific region captured 24.60% of the global market in 2025, generating USD 5.79 billion in revenue, and is projected to reach USD 6.24 billion in 2026.
Latin America
Latin America recorded a market size of USD 1.91 billion in 2025, capturing 8.12% of the global market share, and is projected to reach USD 2 billion in 2026.
Middle East & Africa
In 2025, Middle East & Africa generated USD 1.35 billion, contributing 5.73% to global market revenue, and is projected to grow to USD 1.41 billion in 2026.
Key Market Drivers
"Abbott, Roche Diagnostics, & DiaSorin Expected to Hold Considerable Share in the Global Market "
Giant organizations such as Abbott, Roche Diagnostics, Danaher Corporation, & DiaSorin are expected to lead the market during the forecast period. Optimal research & development strategies, robust supply chain network, increasing investments in research & development, and cost management are projected to serve as huge opportunities for the aforementioned market players to lead the global market during the forecast period. Moreover, a higher potential for intense penetration of immunodiagnostics in the Asia Pacific market may offer significant share for the companies in terms of revenue in the global immunodiagnostics market.
Key companies covered in the report
- Abbott
- Hoffmann-La Roche AG (Roche Diagnostics)
- DiaSorin
- Danaher Corporation
- QIAGEN
- bioMérieux
- Thermo Fisher Scientific
- Quest Diagnostics
Report Overview
At present, developed nations account for the largest immunodiagnostics market share, owing to factors such as growing emphasis on early diagnosis and prevention from diseases by government organizations. Advances in diagnostics testing and changing patient preferences leading to a gradual shift of testing from hospitals and commercial laboratories more towards the point of care is expected to have a higher impact on the growth of the market.
The report provides qualitative and quantitative insights in immunodiagnostics industry and detailed analysis of market size & growth rate for all possible segments in the market. The market is segmented by product type, application, and end user. On the basis of product type, the global market is categorized into instruments and reagents & consumables. Based on the application, the market is segmented into oncology & endocrinology, hepatitis & retrovirus, cardiac markers, infectious diseases, and others.
Various end users covered in the report are hospitals, Clinical laboratories, and physician offices and others. Geographically, the immunodiagnostics market is segmented into five major regions, which are North America, Europe, Asia Pacific, and Latin America, and Middle East & Africa. The regions are further categorized into countries.
Along with this, the report provides elaborative analysis of market dynamics and competitive landscape. Various key insights provided in the report are key brand analysis, key industry developments - mergers, acquisitions and partnerships, reimbursement scenario, and key performance indicators for global immunodiagnostics market
Request for Customization to gain extensive market insights.
Report Scope & Segmentation
|
ATTRIBUTE |
DETAILS |
|
Study Period |
2021-2034 |
|
Base Year |
2025 |
|
Estimated Year |
2026 |
|
Forecast Period |
2026-2034 |
|
Historical Period |
2021-2024 |
|
Unit |
Value (USD Billion) |
|
Growth Rate |
CAGR of 7.58% from 2026 to 2034 |
|
By Product |
· Instruments · Reagents & Consumables |
|
By Application |
· Oncology & Endocrinology · Hepatitis & Retrovirus · Cardiac Markers · Infectious Diseases · Others |
|
By End user
|
· Clinical Laboratories · Hospitals · Physician’s Offices · Others |
|
By Geography |
· North America (USA and Canada) · Europe (UK, Germany, France, Italy, Spain, Scandinavia and Rest of Europe) · Asia Pacific (Japan, China, India, Australia, Southeast Asia and Rest of Asia Pacific) · Latin America (Brazil, Mexico and Rest of Latin America) · Middle East & Africa (South Africa, GCC and Rest of Middle East & Africa) |
Key Industry Developments
- In March 2019, Oncimmune announced the acquisition of Protagen Diagnostics.
- In November 2018, Ortho Clinical Diagnostics received U.S FDA approval for the company’s VITROS HIV Combo test, an immunodiagnostic detect HIV-1 and HIV-2 antibodies (Ab) and the p24 antigen (Ag).
- In May 2016, Diasorin announced the acquisition of Quest Diagnostics’ immunodiagnostics products.
Frequently Asked Questions
As per our (Fortune Business Insights) study, the global immunodiagnostics market is predicted to reach USD 24.99 billion by 2026.
The rise in the geriatric population, technological advancements in the diagnostic kits and rising utilization of immunodiagnostics in rapid point of care testing devices are the key factors driving the immunodiagnostics market.
The reagents and consumables segment is the fastest-growing segment in the immunodiagnostics market by product.
Based on end-users, clinical laboratory segment is the fastest-growing segment during the forecast duration owing to gradual shift of testing from hospitals and clinical laboratories.
North America is anticipated to hold the highest market share in the immunodiagnostics market
Abbott, Roche Diagnostics, and DiaSorin are the top players in the immunodiagnostics market.
The Asia Pacific region is likely to grow at a significant rate in immunodiagnostics market during the forecast attributed to the rising prevalence of infectious diseases, increase in the number of new product approvals, and rising healthcare expenditures
Get 20% Free Customization
Expand Regional and Country Coverage, Segments Analysis, Company Profiles, Competitive Benchmarking, and End-user Insights.
Related Reports
-
US +1 833 909 2966 ( Toll Free )
-
Get In Touch With Us