Short Bowel Syndrome Market Size, Share & Industry Analysis, By Type (GLP-2, Growth Hormone, and Glutamine), By Distribution Channel (Hospital Pharmacies and Online & Retail Pharmacies), and Regional Forecast, 2026-2034
KEY MARKET INSIGHTS
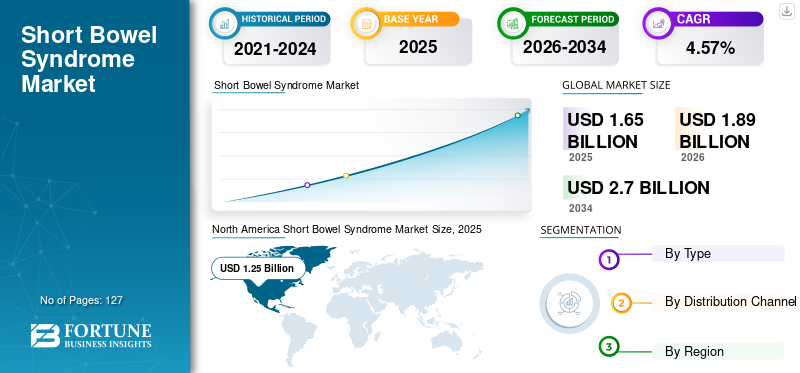
The global short bowel syndrome market size was valued at USD 1.65 billion in 2025. The market is projected to grow from USD 1.89 billion in 2026 to USD 2.7 billion by 2034, exhibiting a CAGR of 4.57% during the forecast period. North America dominated the short bowel syndrome market with a market share of 75.87% in 2025. Moreover, the U.S. short bowel syndrome market size is projected to grow significantly, reaching an estimated value of USD 1.93 billion by 2032, driven by presence of a large patient pool adopting advanced drugs and requiring new treatment options.
There is an increase in the prevalence of short bowel syndrome among the general population globally. Based on various regional and national databases, the condition has affected around 15,000 and 20,000 people in the U.S. and EU-5, respectively. The syndrome is associated with intestinal failure, where the patient pool is highly dependent on parenteral support (PS). There is a rise in the number of patients suffering from short bowel syndrome intestinal failure (SBS-IF), especially in developed countries, owing to factors such as higher diagnosis rates. According to the Crohn's & Colitis Foundation, the number of individuals suffering from short bowel syndrome in the U.S. is approximately 10,000-20,000 people.
Patients suffering from SBS-IF are highly dependent on parenteral support that provides necessary fluids and nutrients. However, comparatively higher costs of treatment and longer duration are factors leading to reduced quality of life of patients. According to the data published by WebMD LLC in 2021, the cost of parenteral support per person is around USD 65,000 to USD 320,000 annually. This, along with the long-term use of parenteral support, impacts the patient’s quality of life and develops various complications such as liver dysfunction, catheter sepsis, and others. This results in the high adoption of drugs such as GLP-2 drugs that offer various advantages, such as increased intestinal absorption and reduced overall parenteral support of the patient.
Currently, there is only one approved GLP-2 drug, Gattex (Revestive), from Takeda Pharmaceutical Company Limited. However, ongoing clinical studies on GLP-2 drugs with more advanced benefits developed by various biotechnology companies are further anticipated to increase the adoption of these drugs during the forecast period. The benefits include improved dosing frequencies, along with clinically proven efficiency of drugs to treat patients with renal failure, among others.
- In September 2022, Zealand Pharma A/S announced the positive results from a phase 3 trial of Glepaglutide for the treatment of short bowel syndrome. Glepaglutide is the potential best-in-class long-acting GLP-2 analog.
Several studies have shown that coronavirus can affect the digestive system, leading to various gastrointestinal complications. The SARS-CoV-2 virus attaches to the angiotensin-converting enzyme 2 (ACE-2) in the intestine, affecting the digestive system.
- According to the data published by UpToDate, Inc., in 2021, it was estimated that one-third of patients who are critically ill from COVID-19 developed gastrointestinal complications.
The SARS-CoV-2 infection caused several complications, such as inflammatory bowel disease and other gastrointestinal tract infections, in conjunction with several other gastrointestinal disorders. Moreover, COVID-19 affected patients with a severe inflammatory bowel disease condition, which resulted in a higher number of cases of Crohn's disease due to a compromised immune system. This led to a rise in the number of patients suffering from SBS.
During the COVID-19 pandemic, the adoption of parenteral nutrition treatment declined due to extended hospital stays and various complications associated with this treatment. This increased the demand for GLP-2-based drugs in the market. All these factors are poised to lead the market growth over the forecast period.
Download Free sample to learn more about this report.
Global Short Bowel Syndrome Market Key Takeaways
Market Size & Forecast:
- 2025 Market Size: USD 1.65 billion
- 2026 Market Size: USD 1.89 billion
- 2034 Forecast Market Size: USD 2.7 billion
- CAGR: 4.57% from 2026 to 2034
Market Share:
- Region: North America dominated the short bowel syndrome market in 2025 with a 75.87% share. This is attributed to a large diagnosed patient pool, increasing adoption of advanced GLP-2 drugs, supportive reimbursement structures, and robust clinical research infrastructure. The U.S. market alone is projected to reach USD 1.93 billion by 2032.
- By Type: The GLP-2 segment held the largest market share in 2026, supported by the presence of the only approved GLP-2 drug, Gattex (Revestive) from Takeda. Pipeline developments and new entrants, such as Glepaglutide (Zealand Pharma) and HM15912 (Hanmi Pharm), are likely to drive this segment further.
Key Country Highlights:
- Japan: Takeda’s Gattex was approved in 2021, marking its entry into the Japanese market. The Ministry of Health, Labor, and Welfare authorized its use, bolstering treatment options for SBS-IF patients and encouraging adoption of GLP-2 analogs.
- United States: The U.S. leads global demand with up to 20,000 SBS patients. High treatment costs (USD 250,000–300,000 per year for Gattex) and complications with parenteral nutrition have driven patients and providers toward GLP-2 therapies. Companies like Ironwood and 9 Meters Biopharma are investing heavily in R&D.
- China & India: Market penetration remains limited due to lack of awareness and limited reimbursement coverage. However, increasing clinical trials, growing rare disease awareness, and expanding distribution networks are creating opportunities for growth.
- Europe: Countries like Germany, France, and the U.K. are increasingly adopting GLP-2 drugs. ESPEN guidelines support GLP-2 as first-line treatment. However, pricing and lower awareness compared to the U.S. result in slower penetration (approx. 3–8% of SBS-IF patients treated with GLP-2 drugs).
Short Bowel Syndrome Market Trends
Shift from Total Parenteral Nutrition to GLP-2 Drugs to Treat SBS
In recent years, the market has witnessed a preferential shift from total parenteral nutrition (TPN) to GLP-2 drugs to treat the disease. This is due to the various complications associated with the long-term usage of parenteral nutrition therapy. The risk factors associated with total parenteral nutrition include liver and kidney dysfunction and catheter sepsis. This results in the adoption of innovative drugs to treat this syndrome that help reduce the overall parenteral support.
The growing adoption of GLP-2 drugs is due to the effective results shown in increasing intestinal absorption, daily once dosage frequency, among others. This, along with the growing penetration rate of GLP-2 medications for this syndrome with intestinal failure, further increases the demand for GLP-2 drugs in the market.
This, along with the ongoing clinical trials on GLP-2-based drugs by key players, is further promoting their adoption in the market. In January 2021, Hanmi Pharm. Co. Ltd’s GLP-2 Analog (HM15912) cleared the phase 2 clinical trial.
The factors mentioned above, the strong focus of key players on receiving various product approvals in the market, and the high cost of parenteral nutrition are responsible for the preferential shift to GLP-2 drugs.
Download Free sample to learn more about this report.
Short Bowel Syndrome Market Growth Factors
Increasing Penetration of GLP-2 Drugs for SBS Intestinal Failure (SBS-IF) to Drive Market Growth
SBS-IF is a rare condition in patients with short bowel syndrome. Patients with this condition require a higher frequency of parenteral support. This treatment is usually administered for an average of 5 days per week. However, long-term use of parenteral support hampers the patient's daily activities, increasing hospitalization rate and complications associated with this treatment, leading to the adoption of GLP-2-based drugs in the market.
In developed countries, such as the U.S., France, Germany, the U.K., and others, the penetration of GLP-2 drugs for the treatment of SBS-IF is greater when compared to the developing economies. Several factors, such as a favorable reimbursement scenario, wide availability of drugs, and higher adoption of GLP-2 drugs among healthcare professionals and patients, have supported the penetration in developed countries.
- Based on few research articles, around 18% to 20% of all patients suffering from SBS-IF (adults and pediatric) are currently being treated by GLP-2 drugs in the U.S. The penetration is slightly lower in Europe (EU-5 countries), where around 3% to 8% of the population suffering from SBS-IF are currently treated by GLP-2 drugs.
- In 2021, Takeda Pharmaceutical Company Limited launched Gattex/Revestive, a subcutaneous injection for short bowel syndrome, in Japan and received approval from the Ministry of Health, Labor and Welfare.
Furthermore, the penetration of GLP-2 drugs is projected to rise during the forecast period in developed countries. This is due to the various benefits offered by pipeline candidates, such as improved dosage frequency and reduced hospitalization rates, among others.
The factors mentioned above, the rising awareness programs to support patients with this syndrome, and the increasing R&D investments by key players in the market are further driving the market growth.
Rising Awareness Programs to Support Patients Suffering from the Syndrome to Boost Market Expansion
The rising incidence of this syndrome in countries such as the U.S., Canada, and the U.K. is increasing the need to develop innovative drugs to treat this condition. According to the data published by Crohn’s & Colitis Foundation, around 10,000 to 20,000 people have this condition in the U.S.
However, the majority of the population from middle and low-income countries are not aware of this condition due to lack of awareness. This shifted the focus of organizations, such as the Oley Foundation, SBS Foundation, and others, to launch awareness programs to support and educate people suffering from this syndrome.
- For example, the SBS foundation provides the SBSCare program, a CE-certified program aiming to improve patient outcomes through medical and nutritional management.
Moreover, the strong focus of key players on research and development investment to develop innovative drugs further propels the market growth.
RESTRAINING FACTORS
High Treatment Cost May Limit Market Growth
Parenteral nutrition is the most commonly used procedure to treat this syndrome, and the overall treatment cost is expensive. This procedure can be done in hospitals and at-home settings. However, the treatment cost is comparatively less for patients receiving home parenteral nutrition than for those who are treated in hospitals.
Moreover, the huge annual cost of the drugs for the treatment of this condition further limits their adoption in the market. For instance, the first approved drug used for treating this syndrome, Gattex/Revestive, is also considered to be expensive.
- According to the data published by NCBI in 2019, it was estimated that the annual cost of Gattex/Revestive drug, used for treating this syndrome, is around USD 250,000 to 300,000 per person annually in Europe. The average cost of Gattex in Asia Pacific is around USD 200,000 to 250,000 per person annually.
In addition, the lack of reimbursement policies and the limited availability of approved drugs in countries such as India, Africa, and others are further limiting the market growth.
Short Bowel Syndrome Market Segmentation Analysis
By Type Analysis
GLP-2 Segment Led Owing to Presence of Approved Products
Based on type, the market has been classified into GLP-2, growth hormone, and glutamine.
The GLP-2 segment led the market accounting for 97.01% market share in 2026. The dominance of the GLP-2 segment is due to a wide range of GLP-2-based pipeline products for SBS. In addition, the presence of approved products in the market further boosts the segment growth.
- For example, in May 2019, the FDA approved Gattex to treat children 1 year and older with SBS. Gattex is a GLP-2 analog drug manufactured by Takeda Pharmaceutical Company Limited.
On the other hand, the growth hormone segment is expected to register a higher CAGR during the forecast period. The growth hormone drugs are approved for short-term use in treating this syndrome. In addition, the presence of approved drugs such as Zorbtive and the insurance drug coverage benefits offered are further expected to fuel the segment growth during the forecast period.
- In July 2022, the pharmacy benefits program covered Merck KGaA’s Zorbtive, a growth hormone used to treat this condition.
To know how our report can help streamline your business, Speak to Analyst
By Distribution Channel Analysis
Preferential Shift of Patients toward Online & Retail Pharmacies to Boost Segment Growth
By distribution channel, the market is classified into hospital pharmacies and online & retail pharmacies.
In 2026, the Online & Retail Pharmacies segment is projected to lead the market with a 60.66% share. The online & retail pharmacies segment is expected to register the highest CAGR during 2024-2032 and generated the highest revenue in 2023. The dominance is attributed to the growing number of online and retail pharmacies in countries such as the U.S., the U.K., India, and others. This, along with the preferential shift of patients toward online platforms, has increased the focus of major players on collaborating with online pharmacies, further propelling the segment growth.
- According to the data published by the Commonwealth Fund in 2021, it was estimated that around 60,000 retail pharmacies are present in the U.S. This provides various opportunities for retail pharmacies to offer innovative drugs for treating this syndrome.
On the other hand, the hospital pharmacies segment is expected to hold a significant share during the assessment period as the rising prevalence of diseases causing SBS, such as Crohn's disease, is increasing the demand for advanced drugs to treat such conditions in the market. The key players focus on providing medicines to hospital pharmacies to meet the increasing demand for these advanced drugs.
REGIONAL INSIGHTS
By region, the market is classified into Europe, North America, Asia Pacific, Latin America, and the Middle East & Africa.
North America
North America Short Bowel Syndrome Market Size, 2025 (USD Billion)
To get more information on the regional analysis of this market, Download Free sample
In 2025, North America generated USD 1.25 billion, contributing 75.87% to global market revenue, and is expected to reach USD 1.45 billion in 2026. The presence of a large patient pool adopting advanced drugs and requiring new treatment options is fueling the short bowel syndrome market growth in this region. In addition, the extended use of approved drugs to treat both adult and pediatric populations, the presence of a wide range of pipeline products, growing R&D activities among the key players, and others is anticipated to contribute to the market growth in this region during the forecast period. The U.S. market is valued at USD 1.43 billion by 2026.
- For instance, Ironwood invested USD 58.2 million in research and development in 2023. The increase is related to the study of Apraglutide in short bowel syndrome, which is designed to support and improve outcomes in patients suffering from short bowel syndrome with intestinal failure (SBS-IF).
Asia Pacific
Asia Pacific recorded a market size of USD 0.17 billion in 2025, capturing 10.03% of the global market share, and is expected to reach USD 0.19 billion in 2026. The direct presence of major players in the market, huge R&D investments in developing innovative drugs, and the rising focus on clinical trials of GLP-2-based drugs propel the market growth in this region. In addition, the rising awareness of rare diseases in countries such as India, Japan, Australia, and others is resulting in the growing demand and need for various treatment options in this region. The Japan market is valued at USD 0.14 billion by 2026, the China market is valued at USD 0.03 billion by 2026
- In 2021, Takeda Pharmaceutical Company Limited announced the approval for marketing and distribution of its GLP-2 drug, Gattex, from the Ministry of Health, Labor, and Welfare, Japan.
Europe
The Europe market accounted for USD 0.18 billion in 2025, representing 11.16% of the global industry, and is expected to reach USD 0.19 billion in 2026. Europe held the second position in the global market. The U.K. market continues to grow, projected to reach a market value of USD 0.03 billion in 2025. The direct presence and emergence of new clinical-stage biotechnology companies in this region, mainly focusing on developing innovative drugs and new biologics for treating rare diseases, is fueling European market growth. This, along with the various guidelines laid to support patients suffering from this condition is stimulating market growth. The UK market is valued at USD 0.03 billion by 2026, while the Germany market is valued at USD 0.07 billion by 2026.
- For example, according to the European Society for Clinical Nutrition and Metabolism (ESPEN) guidelines, if the growth factor treatment is taken into account for treating this condition, GLP-2 analog is considered the first line of treatment.
Middle East & Africa and Latin America
The Middle East & Africa market generated USD 0.03 billion in 2025, representing 1.56% of the global market landscape, and is expected to reach USD 0.03 billion in 2026. Latin America accounted for USD 0.02 billion in 2025, representing 1.38% of the global market share, and is projected to reach USD 0.02 billion in 2026. The rising awareness of rare diseases and the growing need and demand for advanced products in these regions are poised to lead to growth during the forecast period. In addition, key players are focusing on expanding their distribution network in these regions to meet the growing demand, further fueling the market growth. The UAE market is foreseen to stand at USD 0.01 billion in 2025.
Rest Of World
In July 2020, Nestlé Health Science invested USD 50.0 million to establish a manufacturing center in the U.S., which manufactures infant formulas and medical nutrition products such as peptamen. This investment also focuses on distributing the products in Canada, Brazil, Mexico, Australia, and Taiwan.
KEY INDUSTRY PLAYERS
Leading Players Focus on Robust Product and Pipeline Portfolio to Enhance Their Position
In terms of the competitive landscape, the global market is dominated by Takeda Pharmaceutical Company Limited, which accounted for the highest short bowel syndrome market share in 2023. The company’s strong focus on R&D investments for developing and delivering new drugs in the market and entering into mergers and acquisitions with other players to expand its presence is responsible for its highest share in the market.
- In May 2024, Takeda Pharmaceutical Company Limited in collaboration with Inspire presented new research at Digestive Disease Week (DDW) 2024, and unveiled mixed-method study on Short Bowel Syndrome (SBS) patient experience. This study was focusing on physical, social, and emotional burdens faced by SBS patients and their caregivers.
In addition, new clinical-stage companies such as VectivBio AG, 9 Meters Biopharma, Inc., Zealand Pharma A/S, and others primarily focus on developing advanced and personalized drugs for treating rare diseases such as short bowel syndrome and graft versus host disease. Moreover, the presence of pipeline products with rising number of clinical studies is likely to support the growing share of the market players globally.
- In March 2022, VectivBio AG announced a licensing agreement with Asahi Kasei Pharma Corporation to commercialize and develop apraglutide in Japan, which is used to treat SBS.
List of Top Short Bowel Syndrome Companies:
- Takeda Pharmaceutical Company Limited (Japan)
- OxThera (Sweden)
- ZEALAND PHARMA (Denmark)
- Ironwood (U.S.)
- Hanmi Pharm Co., Ltd. (South Korea)
- Jaguar Health (U.S.)
- Merck KGaA (Germany)
- EnteraBio Ltd. (Israel)
- Nestlé (Switzerland)
KEY INDUSTRY DEVELOPMENTS:
- September 2023- EnteraBio Ltd. entered into a research collaboration agreement with OPKO Biologics, Inc. under the collaboration, OPKO supplied its proprietary long-acting GLP-2 peptide and Oxyntomodulin (OXM) analogs for the development of oral tablet formulations for obesity and intestinal malabsorption syndromes using Entera's proprietary oral delivery technology.
- March 2023 – Takeda Pharmaceutical Company Limited participated in the 18th European Crohn’s and Colitis Organisation (ECCO) Congress. The company shared practical tips for increasing treatment adherence, discussed new attitudes to treatment goals, and showcased real clinical experience for the disease.
- June 2022 – 9 Meters Biopharma, Inc. announced the positive results of the phase 2 clinical study of Vurolenatide in treating this syndrome.
- August 2021 – Zealand Pharma A/S announced the dose administration of Glepaglutide to the first patient in the phase 3b clinical trial for the treatment of short bowel syndrome.
- April 2021 – Hanmi Pharm.Co., Ltd. announced that it has received fast-track designation by the U.S. FDA for its HM15912(GLP-2 Analog) used for treating this syndrome.
REPORT COVERAGE
The global market research report provides a detailed analysis of the market. It focuses on key aspects such as leading companies, types, and distribution channels. Besides, it offers insights into the market trends and highlights key industry developments. In addition to the factors mentioned above, the report encompasses several factors that have contributed to the market's growth in recent years.
Request for Customization to gain extensive market insights.
Report Scope & Segmentation
|
ATTRIBUTE |
DETAILS |
|
Study Period |
2021-2034 |
|
Base Year |
2025 |
|
Estimated Year |
2026 |
|
Forecast Period |
2026-2034 |
|
Historical Period |
2021-2024 |
|
Growth Rate |
CAGR of 4.57% from 2026-2034 |
|
Unit |
Value (USD billion) |
|
Segmentation |
By Type
|
|
By Distribution Channel
|
|
|
By Region
|
Frequently Asked Questions
Fortune Business Insights says that the market is projected to reach USD 2.7 billion by 2034.
In 2025, the market stood at USD 1.65 billion.
The market is expected to exhibit steady growth at a CAGR of 4.57% during the forecast period.
By type, the GLP-2 segment led in 2026.
The increasing penetration of GLP-2 drugs among patients suffering from SBS-IF globally, coupled with awareness programs by various organizations, is set to drive the market growth.
Takeda Pharmaceutical Company, Merck KGaA, and Emmaus Medical, Inc. are the top players in the market.
North America is expected to hold the highest market share in the market.
Get 20% Free Customization
Expand Regional and Country Coverage, Segments Analysis, Company Profiles, Competitive Benchmarking, and End-user Insights.
Related Reports
-
US +1 833 909 2966 ( Toll Free )
-
Get In Touch With Us