U.S. Immunoglobulin Market Size, Share & Industry Analysis, By Product Type (Intravenous Immunoglobulin (IVIG) {Primary Immunodeficiency, Secondary Immunodeficiency, Chronic Inflammatory Demyelinating Polyneuropathy, Guillain-Barré Syndrome, Immune Thrombocytopenic Purpura, Multifocal Motor Neuropathy, and Others}, and Subcutaneous Immunoglobulin (SCIG) {Primary Immunodeficiency, Secondary Immunodeficiency, Chronic Inflammatory Demyelinating Polyneuropathy, and Others}), By Form (Liquid and Lyophilized), By End User (Hospitals, Clinics, and Homecare), and Country Forecast, 2026-2034
U.S. Immunoglobulin Market Size and Future Outlook
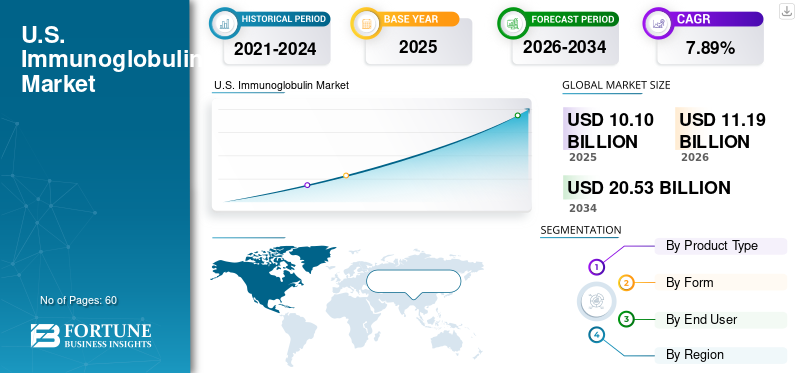
The U.S. immunoglobulin market size was valued at USD 10.10 billion in 2025. The market is projected to grow from USD 11.19 billion in 2026 to USD 20.53 billion by 2034, exhibiting a CAGR of 7.89% during the forecast period.
The U.S. market growth is driven by the novel immunoglobulin product launches by key companies in the country to cater to the increasing demand of patients with immunodeficiency. Robust healthcare infrastructure, favorable government support, and the country's research ecosystem, which assist in developing and expanding the immunoglobulin clinical pipeline, further strengthen market growth.
- For instance, in September 2024, GC Biopharma USA, Inc. launched its immunoglobulin (IG) product ALYGLO (immune globulin intravenous, human-stwk), the company's first 10% intravenous immunoglobulin therapy for the treatment of adult patients aged 17 years and older with primary humoral immunodeficiency (PI) in the U.S.
Moreover, key players in the immunoglobulin treatment industry, such as CSL, Octapharma AG, and Takeda Pharmaceutical Company Limited, are expanding their product offerings to strengthen their market positions.
Download Free sample to learn more about this report.
U.S. IMMUNOGLOBULIN MARKET TRENDS
Integration of Infusion Devices for Subcutaneous Administration is a Prominent Trend
A major U.S. market trend observed is the integration of devices for facilitated subcutaneous administration in the home care setting. These new devices make it simpler and less intimidating to administer immunoglobulin at home. They reduce steps such as mixing, drawing up doses, and handling multiple supplies, helping patients self-administer more. As a result, companies are investing in user-friendly devices to make at-home SCIG easier, thereby increasing adoption and supporting market growth.
- For instance, in October 2025, Takeda Pharmaceutical Company Limited received approval from the U.S. FDA for HyHub and HyHub Duo devices available in the U.S. for patients 17 years of age and older. The dedicated devices facilitate subcutaneous immunoglobulin (SCIg) infusion using dual vial units (DVUs) of immunoglobulin and hyaluronidase, allowing transfer from vials without a needle or pooling bag in a home environment or clinical setting.
Download Free sample to learn more about this report.
MARKET DYNAMICS
MARKET DRIVERS
Increasing Research and Development to Fuel Immunoglobulin Market Growth
One of the principal factors driving the immunoglobulin market's growth is the U.S.'s increasing research and development initiatives by key companies for new product launches and expanded manufacturing capabilities. When companies invest in R&D, they can improve efficiency in immunoglobulin production, develop better formulations, and generate stronger clinical evidence for broader use. This helps a company get more output from the same plasma or run production more reliably, which improves supply and eases shortages, directly increasing overall demand and market growth.
- For instance, in April 2025, ADMA Biologics, Inc. received U.S. FDA approval for its innovative yield-enhancement production process. This innovative process has demonstrated an ability to increase production yields by approximately 20% from the same starting plasma volume.
MARKET RESTRAINTS
Strict Quality and Safety Oversight to Limit Adoption of Products
As immunoglobulins are derived from plasma, they are subject to strict quality and safety oversight in the U.S. When regulators or manufacturers identify a potential safety signal, lots can be withdrawn, and companies must investigate and fix the issue. This creates a disruption in the supply chain, affecting treatment plans in hospitals and resulting in delays. When patients face delays or switches, overall market growth slows, adversely impacting market growth.
- For instance, in April 2025, ADMA Biologics Inc. initiated a voluntary withdrawal of one lot of BIVIGAM, 10% of 10g vial, due to the potential for an increased rate of allergic/hypersensitivity reactions. Such instances erode consumer trust.
MARKET OPPORTUNITIES
Expanding Plasma Collection to Offer Significant Opportunity for Market Players
One significant challenge for the immunoglobulin market is supply chain disruptions caused by limited plasma availability and low production capacity among key players. Thus, improving plasma collection capacity to meet the increasing demand for immunoglobulins creates a major growth opportunity for the market. Increasing investments by key players for expansion of plasma collection centers offer significant growth opportunities.
- For instance, in September 2024, Kamada Ltd. expanded its plasma collection operations with the opening of a new plasma collection center in Houston, Texas. The new 12,000 square foot center is operated by Kamada’s wholly owned subsidiary, Kamada Plasma, and is planned to support over 50 donor beds with an estimated total collection capacity of approximately 50,000 liters annually.
MARKET CHALLENGES
Manufacturing and Maintenance Issues Triggering Shortages Pose a Critical Challenge to Market Growth
Immunoglobulins are complex biomolecules and require critical production requirements. Even small production issues can reduce supply drastically. When manufacturers face manufacturing issues, plant maintenance requirements, or ingredient shortages, the result is often shortage status. That causes shortage of products results in delayed treatment and slower market growth.
- For instance, in November 2025, Kedrion Spa reported shortage of Rho(D) Immune Globulin (Human) due to the limited availability of an active ingredient and manufacturing issues and plant maintenance requirements. These factors affect the supply chain of the immunoglobulin and adversely impacts its growth potential.
Segmentation Analysis
By Product Type
High Utilization of IVIG to Position Them in Leading Position and Boost Segmental Growth
Based on product type, the market is categorized into intravenous immunoglobulin (IVIG) and subcutaneous immunoglobulin (SCIG). The intravenous immunoglobulin (IVIG) segment is further sub-segmented into primary immunodeficiency, secondary immunodeficiency, chronic inflammatory demyelinating polyneuropathy, Guillain-Barré syndrome, immune thrombocytopenic purpura, multifocal motor neuropathy, and others. The subcutaneous immunoglobulin (SCIG) segment is further divided into primary immunodeficiency, secondary immunodeficiency, chronic inflammatory demyelinating polyneuropathy, and others.
Among these, the intravenous immunoglobulin (IVIG) segment dominated the U.S. immunoglobulin market share. The segment's dominance is attributed to high utilization and broad clinical familiarity of IVIG immunoglobulins. Due to healthcare providers' preference for IVIG immunoglobulins, key companies are focusing on new product launches and subsequent approvals from relevant regulatory bodies.
- In February 2021, Pfizer Inc. received a supplemental Biologics License Application (sBLA) from the U.S. FDA for PANZYGA to treat adult patients with chronic inflammatory demyelinating polyneuropathy (CIDP), a rare peripheral nerve disorder.
The subcutaneous immunoglobulin (SCIG) segment is expected to grow at the fastest CAGR of 11.02% over the U.S. immunoglobulin market forecast period.
To know how our report can help streamline your business, Speak to Analyst
By Form
Increasing Use of Ready-to-Use Format of Liquid Immunoglobulin to Lead Segmental Growth
Based on form, the market is classified into liquid and lyophilized.
In 2025, the liquid segment accounted for the largest share in the country, with a significant CAGR. The ready-to-use liquid immunoglobulin format facilitates administration. They are also a preferred alternative for replacement therapy. Underscoring these advantages, key companies are focusing their resources on new product launches, driving the segmental growth. Additionally, government support and prompt regulatory approval support the market's growth.
- For instance, in June 2025, Takeda Pharmaceutical Company Limited received approval from the U.S. FDA for GAMMAGARD LIQUID ERC [immune globulin infusion (human)] with in a 10% solution, the only ready-to-use liquid immunoglobulin (IG) therapy with low immunoglobulin A (IgA) content, as replacement therapy for people two years of age and older with primary immunodeficiency (PI). Such initiatives are contributing to the segment's growth.
The lyophilized segment is projected to grow at a CAGR of 2.74% during the forecast period for the U.S. market.
By End User
Preference toward Hospital Settings by Healthcare Providers to Place Them in a Leading Position
Based on end user, the market is segmented into hospitals, clinics, and home care.
In terms of end users, the hospital segment dominated the U.S. market in 2025. It accounted for the largest market share due to high demand for the treatment of various immunodeficiency diseases. They are the first point of contact for accessing healthcare services. Furthermore, strategic collaborations among hospitals and other key operating entities to raise awareness further reinforce the strong market share.
- For instance, in April 2025, the Jeffrey Modell Foundation participated in World Primary Immunodeficiency (PI) Week, held annually from April 22 to 29, to raise awareness of primary Immunodeficiencies, and provide early access to care for patients and families worldwide. Such initiatives raise awareness and boost the adoption of immunotherapies, fueling U.S. immunoglobulin market growth.
The home care segment is projected to grow at a CAGR of 8.45% during the study period.
COMPETITIVE LANDSCAPE
Key Industry Players
Focus on New Product Launches by Key Players to Propel Market Progress
The U.S. market is highly consolidated, with companies such as Octapharma AG, Grifols, S.A., Takeda Pharmaceutical Company Limited, and CSL holding significant market share. Strategic partnerships, new product launches, regulatory approvals, and increasing investments in the sector drive these companies' market share.
- For instance, in October 2025, Grifols, S.A. launched Yimmugo in the U.S. for the treatment of primary Immunodeficiencies (PID). The treatment was produced in collaboration with Biotest, a Grifols Group company, such developments aimed to drive market growth.
Other notable players in the U.S. market include Kedrion S.p.A. and Johnson & Johnson. These companies are expected to prioritize expanding manufacturing capacity, strategic collaborations, and new product launches to strengthen their position during the forecast period for the U.S. market.
LIST OF KEY U.S. IMMUNOGLOBULIN COMPANIES PROFILED
- CSL (Australia)
- Takeda Pharmaceutical Company Limited (Japan)
- Grifols, S.A. (Spain)
- Kedrion S.p.A (Italy)
- Octapharma AG (Switzerland)
- ADMA Biologics, Inc. (U.S.)
- Taibang Biologic Group (China Biologic Products Holdings, Inc.) (China)
- LFB Group (France)
- Shanghai RAAS Blood Products Co., Ltd (China)
- GC Biopharma Corp (South Korea)
KEY INDUSTRY DEVELOPMENTS
- September 2025: Kedrion S.p.A. received approval from the U.S. FDA for QIVIGY, a new 10% Immunoglobulin for intravenous use, indicated for treatment of adults with Primary humoral Immunodeficiency (PI).
- May 2025: Grifols, S.A. submitted an Investigational New Drug (IND) application to the U.S. FDA to initiate a Phase 2 trial evaluating its immunoglobulin (IG) drops, GRF312 Ophthalmic Solution, as a potential new treatment for dry eye disease (DED).
- September 2024: GC Biopharma launched ALYGLO (Immune Globulin Intravenous, Human-stwk) 10% liquid for adults, an immune globulin for the treatment of primary humoral immunodeficiency (PI) in the U.S.
- March 2024: ADMA Biologics, Inc. announced the U.S. Food and Drug Administration’s (FDA) approval for its supplemental Biologics License Applications (BLAs) for both ASCENIV and BIVIGAM to extend the approved 4-week room temperature (25°C) storage conditions during the first 24 months of shelf life, to allow for a 4-week room temperature storage at any time during the entire 36-month approved shelf life
- January 2023: Eisai Co., Ltd. Collaborated with Biogen Inc. and received Accelerated Approval Pathway from the U.S. FDA for lecanemab-irmb (LEQEMBI) 100 mg/mL injection for intravenous use, a humanized immunoglobulin gamma 1 (IgG1) monoclonal antibody directed for the treatment of Alzheimer’s disease (AD).
REPORT COVERAGE
Request for Customization to gain extensive market insights.
Report Scope & Segmentation
|
ATTRIBUTE |
DETAILS |
|
Study Period |
2021-2034 |
|
Base Year |
2025 |
|
Estimated Year |
2026 |
|
Forecast Period |
2026-2034 |
|
Historical Period |
2021-2024 |
|
Growth Rate |
CAGR of 7.89% from 2026 to 2034 |
|
Unit |
Value (USD Billion) |
|
Segmentation |
By Product Type, Form, End User, and Region |
|
By Product Type |
· Intravenous Immunoglobulin (IVIG) o Primary Immunodeficiency o Secondary Immunodeficiency o Chronic Inflammatory Demyelinating Polyneuropathy o Guillain-Barré Syndrome o Immune Thrombocytopenic Purpura o Multifocal Motor Neuropathy o Others · Subcutaneous Immunoglobulin (SCIG) o Primary Immunodeficiency o Secondary Immunodeficiency o Chronic Inflammatory Demyelinating Polyneuropathy o Others |
|
By Form |
· Liquid · Lyophilized |
|
By End User |
· Hospitals · Clinics · Homecare |
Frequently Asked Questions
In 2025, the market value stood at USD 10.10 billion.
The market is expected to grow at a CAGR of 7.89% over the forecast period.
By product type, the intravenous immunoglobulin (IVIG) segment is expected to lead the market.
The rising prevalence of immunodeficiency diseases is driving demand for immunoglobulins in the U.S. and boosting market growth.
Octapharma AG, Takeda Pharmaceutical Company Limited, and CSL are the major players in the U.S. market.
Get 20% Free Customization
Expand Regional and Country Coverage, Segments Analysis, Company Profiles, Competitive Benchmarking, and End-user Insights.
Related Reports
-
US +1 833 909 2966 ( Toll Free )
-
Get In Touch With Us