Orphan Drugs Market Size, Share & Industry Analysis, By Therapy Area (Oncology, Hematology, Neurology, Endocrinology, Cardiovascular, Respiratory, Immunotherapy, Infectious Diseases, and Others), By Drug Type (Biologics and Non-Biologics), By Distribution Channel (Hospital Pharmacy, Retail Pharmacy, Online Pharmacy, and Others), and Regional Forecast, 2026-2034
KEY MARKET INSIGHTS
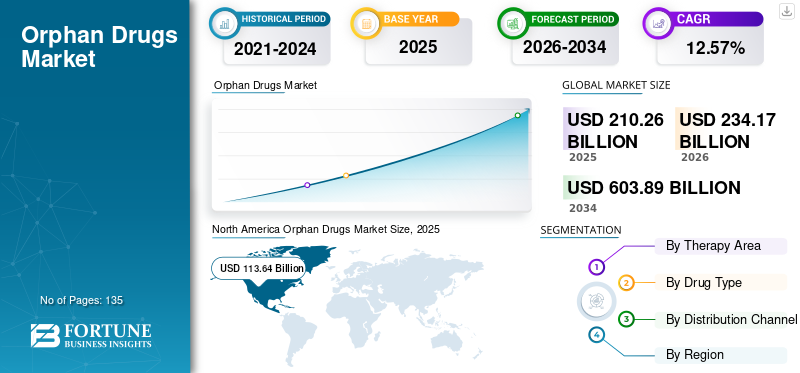
The global orphan drugs market size was valued at USD 210.26 billion in 2025 and is projected to grow from USD 234.17 billion in 2026 to USD 603.89 billion by 2034, exhibiting a CAGR of 12.57% during the forecast period. North America dominated the global market with a share of 54.04% in 2025. Moreover, the U.S. orphan drugs market size market size is projected to grow significantly, reaching an estimated value of USD 237.39 billion by 2032, driven by favorable government policies for orpahn drugs.
Orphan drugs are developed by the pharmaceutical industry for economic reasons but respond to the public health needs. These drugs are developed to treat patients suffering from serious diseases for which no treatment, or at least a satisfactory one, has so far been available. For instance, an article published by the Orphanet in 2023 stated that the number of rare diseases for which no treatment is currently available is estimated to be between 4,000 and 5,000 globally. Moreover, 25 to 30 million people are reported to be affected by these rare diseases in Europe.
According to the European Organization for Rare Diseases, orphan drugs are produced for diagnosis, prevention, and treatment of rare disorders. The oncology therapy area holds a considerably higher share in the global market due to emergence of new cancer-related diseases. Additionally, FDA approvals to a series of cancer-related orphan drugs are expected to result in increased commercialization of these drugs, especially in the oncology therapy area, during the forecast period. The market will derive growth from higher demand for immunomodulators, as they regularize or amplify the immune system. Rising prevalence of infectious diseases in developed as well as emerging nations and higher prices of medications are projected to fuel the demand for immunomodulators, thus contributing to the global orphan drugs market growth during the forecast period.
The COVID-19 pandemic had a positive impact on the market growth due to an increase in regulatory approvals. In 2021, the growth of the market continued primarily due to the resumption of research and development activities of market players. This growth is expected to continue during the forecast period, owing to the launch of various pipeline candidates for various indications by key players. Such trends indicate that the market is poised for steady growth during the forecast period.
Download Free sample to learn more about this report.
Orphan Drugs Market Snapshot & Highlights
Market Size & Forecast:
- 2025 Market Size: USD 210.26 billion
- 2026 Market Size: USD 234.17 billion
- 2034 Forecast Market Size: USD 603.89 billion
- CAGR: 12.57% from 2026–2034
Market Share:
- North America dominated the orphan drugs market in 2025 with a 54.04% share, driven by favorable regulatory frameworks, significant R&D investments, and a large patient population with rare diseases. The U.S. market alone is projected to reach USD 237.39 billion by 2032, supported by government incentives and high treatment demand.
- By Therapy Area, oncology held the largest share in 2023 due to the high number of cancer-related orphan drug approvals and the presence of robust development pipelines. FDA approvals, such as Pacritinib for myelofibrosis, continue to fuel this segment.
Key Country Highlights:
- Japan: Growth is driven by increasing awareness of rare diseases, supportive regulatory reforms, and national strategies promoting precision medicine and gene therapies.
- United States: Strong growth is expected due to the large patient population (approx. 30 million Americans with rare diseases), advanced R&D ecosystem, favorable orphan drug policies (e.g., market exclusivity, tax credits), and numerous FDA approvals.
- China: Rising healthcare spending, policy support for rare diseases, and increasing participation of domestic biotech firms are contributing to market expansion.
- Europe: With over 30 million people affected by rare diseases, initiatives like the EU Orphan Regulation and a growing pipeline of drugs are aiding market growth.
Orphan Drugs Market Trends
Strong Product Launches to Drive Market Growth
One of the prominent trends in the global market is strong product launches for these disease indications. Globally, patients suffering from rare diseases are seeking efficient and potent treatment for these disorders. Increasing R&D initiatives and rising advocacy by patients’ groups for effective treatment are propelling the introduction of newer and more effective product offerings. These trends have led to the development and marketing of several blockbuster drugs for the treatment of many types of cancers, immunological diseases, and other rare disorders.
- As per the Orphan Drug Report 2022 by Evaluate Pharma, it is estimated that the rise of top 10 orphan drugs blockbusters will be worth between USD 3.0 billion and USD 13 billion by 2026. Furthermore, it estimated that 40% of Johnson & Johnson’s pharmaceutical sales will come from orphan drugs, especially through blood cancer drugs, such as Imbruvica and Darzalex, which is expected to be worth over USD 23.0 billion by 2026. Thus, the introduction of such sophisticated orphan therapeutics by established and emerging companies have been instrumental in boosting the adoption of these drugs, which is expected to aid the growth of the global market during the forecast period.
Download Free sample to learn more about this report.
Orphan Drugs Market Growth Factors
Increasing R&D Initiatives to Drive Market Growth
One of the critical driving factors prevailing in the global market is the increasing R&D investments by prominent players for the development of novel products. Since the awareness and understanding regarding rare diseases has increased, a number of key clinical stage biopharmaceutical companies and established market players are creating strong pipeline candidates for orphan drugs in various stages of clinical trials. This increasing foray into rare disorder therapeutics is due to the reason that major pharmaceutical breakthroughs resulting in the development of blockbuster drugs are quite possible in rare disorders in comparison to the traditional pharmaceutical portfolios.
- In 2021, over half of FDA’s Center for Drug Evaluation and Research (CDER) approvals had orphan designation and two CAR-T cell therapies were approved by the Center for Biologics Evaluation and Research (CBER).
Similarly, government bodies of some countries are undertaking initiatives to promote research and initiative activities. Such factors are expected to drive the market growth over the forecast period.
- In October 2023, the Indian government launched the “National Policy on Research and Development and Innovation in Pharma-MedTech Sector in India” and the “Scheme for Promotion of Research and Innovation in Pharma MedTech Sector (PRIP)”. They focus on fostering research, development, and innovation in the pharmaceutical and medical technology sectors in India. The policy’s parts focus on rare diseases, gene therapy, orphan drugs, and precision medicines.
Another strong driver for this is that pharmaceutical companies are required to conduct larger outcome studies for the regulatory approvals of traditional therapeutics for diseases, such as diabetes and Coronary Artery Disease (CAD) in comparison to rare diseases. This factor is projected to propel the market growth during the forecast period.
Growing Patient Population with Rare Diseases is Propelling Market Development
The patient population with rare diseases has been growing across the globe. This substantial patient pool has increased the demand for suitable treatment options. Such scenarios have also augmented strong research and development initiatives and product launches for rare disease therapeutics. These factors are expected to have a significant positive impact on the global market.
- For intance, according to an article published by Euronews in 2023, there are 5,000 to 8,000 rare diseases in Europe and affects the daily life of almost 30.0 million people in the region and 300.0 million across the world. This has created a sizable patient population who have significantly unmet clinical needs, which are extremely serious in nature. Hence, the growing patient population with rare diseases and the increasing involvement of major and emerging market players in the development of sophisticated and effective products are expected to boost market growth.
RESTRAINING FACTORS
High Cost Associated with Orphan Drugs to Limit their Adoption
Despite the increasing focus on the development and marketing of rare disease therapeutics globally, certain limitations are restricting the growth of the global market. One of the critical restraining factors is the high costs associated with these drugs.
- As per a news article published by USA Today in 2023, Zolgensma, a one-time treatment approved for treating spinal muscular atrophy, is among the highest-priced drugs in the U.S. market. The drugs cost around USD 2.25 million and have treated more than 3,000 patients globally.
In developing countries, the drugs may not be adequately reimbursed, significantly increasing the patients' out-of-pocket costs. This creates a cost burden for treatment over the patients, which results in the limited adoption of these products. Such scenarios limit the market growth.
Orphan Drugs Market Segmentation Analysis
By Therapy Area Analysis
Oncology Segment to Dominate due to Robust Regulatory Drug Approvals
On the basis of therapy area, the market is classified into oncology, hematology, neurology, endocrinology, cardiovascular, respiratory, immunotherapy, infectious diseases, and others.
The oncology segment is projected to dominate the market with a share of 47.42% in 2026. The dominance is due to the presence of several oncology drugs in the product development pipelines of key players and presence of a large number of orphan drugs dedicated to the treatment of various cancers. For instance, in February 2022, CTI BioPharma received the U.S. FDA approval for its drug Pacritinib for the treatment of adult patients with myelofibrosis, a rare bone cancer affecting over 21,000 patients in the U.S.
The hematology segment is projected to be the second most dominant segment owing to a number of new product launches and increasing number of regulatory approvals. The neurology segment is also anticipated to register a comparatively strong CAGR due to positive developments in the product offerings for chronic diseases, such as multiple sclerosis.
However, the endocrinology, respiratory, and cardiovascular segments are anticipated to register lower CAGRs during the forecast period. The immunotherapy segment is expected to register a strong CAGR due to the presence of reliable products, such as Keytruda, and also increased research initiatives.
To know how our report can help streamline your business, Speak to Analyst
By Drug Type Analysis
Biologics Segment Dominated Due to Strong Product Offerings
Based on drug type, the market is segmented into biologics and non-biologics.
The biologics segment is projected to dominate the market with a share of 74.44% in 2026 due to the overwhelming presence of product offerings classified as biologics. According to an estimate by the U.S. Food and Drug Administration (FDA), the Office of Orphan Products Development (OOPD) of the organization has developed and marketed over 600 drugs and biologic products to treat rare disorders since 1983. The non-biologics segment accounted for a lower share of the global market and also registered a comparatively low CAGR.
By Distribution Channel Analysis
High Accessibility toward Hospital Pharmacies Led to Segment Dominance
On the basis of distribution channel, the market is segmented into hospital pharmacy, retail pharmacy, online pharmacy, and others.
The hospital pharmacy segment is projected to dominate the market with a share of 46.42% in 2026. The primary reason behind the segment’s dominance is that a significantly large number of drugs have to be administered at hospitals intravenously by trained healthcare professionals. The retail pharmacy segment is expected to register a comparatively lower CAGR during the forecast period.
The online pharmacy segment is expected to register a high CAGR due to the ease given to consumers while buying drugs. Also, during the COVID-19 pandemic, online pharmacies registered strong growth due to the increasing number of consumers switching to online pharmacies to purchase these drugs.
REGIONAL INSIGHTS
In terms of region, the global market is segmented into North America, Europe, Asia Pacific, and the Rest of the World.
North America
North America Orphan Drugs Market Size, 2025 (USD Billion)
To get more information on the regional analysis of this market, Download Free sample
The North America market was valued at USD 113.64 billion in 2025, capturing 54.04% of global revenue, and is estimated to reach USD 126.67 billion in 2026. The dominance of North America is attributed to the significant expenditure on orphan drugs, strong patient population, and presence of key market players leading to the development of sophisticated and innovative products. The U.S. market is projected to reach USD 117.04 billion by 2026.
- For instance, according to the Genetic and Rare Diseases (GARD) Information Center, there are more than 10,000 known rare diseases in the U.S. that affect about 1 in 10 people or 30 million people in the U.S. This factor, coupled with the presence of favorable reimbursement policies in the U.S., is responsible for North America’s dominance in the global market.
- In December 2023, BioVersys AG received the U.S. Food and Drug Administration grant of orphan-drug designation (ODD) for alpibectir (BVL-GSK098) and ethionamide fixed-dose combination. It is used for the treatment of tuberculosis (TB).
Europe
In 2025, Europe held 25.66% of the global market, reaching a valuation of USD 53.94 billion, and is projected to grow to USD 59.93 billion in 2026, owing to the rising patient population in the region and strong adoption of sophisticated rare disease therapeutics. The UK market is projected to reach USD 8.17 billion by 2026, and the Germany market is projected to reach USD 12.5 billion by 2026. In 2025, Europe held 25.66% of the global market, reaching a valuation of USD 53.94 billion, and is projected to grow to USD 59.93 billion in 2026.
Asia Pacific
The market in Asia Pacific reached USD 35.76 billion in 2025, representing 17.00% of total market revenue, and is projected to reach USD 40.08 billion in 2026, owing to the increasing healthcare expenditure and high awareness of rare diseases. The Japan market is projected to reach USD 11.5 billion by 2026, the China market is projected to reach USD 8.26 billion by 2026, and the India market is projected to reach USD 5.83 billion by 2026. The market in Asia Pacific reached USD 35.76 billion in 2025, representing 17.00% of total market revenue, and is projected to reach USD 40.08 billion in 2026.
Latin America and Middle East & Africa
Latin America and the Middle East & Africa, which accounted for a lower share of the global market owing to the under penetration of these medicines in the regions. Latin America maintained a strong presence in the global market, reaching USD NA billion in 2025, accounting for NA share, and is expected to reach USD NA billion in 2026.
Rest of the World
In 2025, Rest of the World generated USD 6.93 billion, contributing 3.29% to global market revenue, and is projected to grow to USD 7.48 billion in 2026.
List of Key Companies in Orphan Drugs Market
Strong Portfolio of Bristol-Myers Squibb Company and F. Hoffmann-La Roche Ltd. to Drive Market Growth
The market is highly competitive in nature, owing to the presence of a large number of key market players and their diverse product development pipelines. Currently, Bristol-Myers Squibb Company and F. Hoffmann La Roche Ltd. are dominating the market, accounting for a significant share of the global market in 2023. This is primarily due to the strong sales and product portfolios of oncology-related drugs of these companies.
However, several key players, such as Biogen Inc. and BioMarin Pharmaceutical Inc., with their innovative product pipelines for various rare diseases, are emerging in this industry. Other prominent players include Amgen, Alexion, Novartis, AstraZeneca, DAIICHI SANKYO COMPANY LIMITED, and Agios Pharmaceuticals who also have a significant presence in the global market of novel drugs.
LIST OF KEY COMPANIES PROFILED:
- Amgen Inc. (U.S.)
- Bayer AG (Germany)
- F. Hoffmann-La Roche Ltd (Switzerland)
- Alexion Pharmaceuticals Inc. (U.S.)
- Novo Nordisk A/S (Denmark)
- Novartis AG (Switzerland)
- Bristol-Myers Squibb Company (U.S.)
- AstraZeneca (U.K.)
- DAIICHI SANKYO COMPANY, LIMITED (Japan)
- GlaxoSmithKline plc (U.K.)
KEY INDUSTRY DEVELOPMENTS:
- February 2024 – Rapid Commercialization Partners (RCP) partnered with Orphan Now to help companies developing orphan drugs accelerate time to market and get treatments to patients around the world more efficiently.
- February 2023 – GSK plc’s Benlysta (belimumab) received the U.S. Food and Drug Administration's (FDA) Orphan Drug Designation (ODD). It is a B-cell inhibiting monoclonal antibody to treat systemic sclerosis. GSK plans to initiate a phase II/III trial of belimumab for systemic sclerosis-associated interstitial lung disease (SSc-ILD) in the first half of 2023.
- December 2022 – Amgen Inc. announced its decision to acquire Horizon Therapeutics Plc at a value of USD 27.8 billion along with its orphan drug portfolio.
- November 2022 – Alexion, a group of AstraZeneca focused on rare diseases, acquired LogicBio Therapeutics, Inc., a pioneering genomic medicine company. This acquisition was made to accelerate Alexion’s growth in genomic medicine while treating rare diseases.
- March 2022 – Marinus Pharmaceuticals received the U.S. FDA approval for Ztalmy (ganaxolone) that treats seizures associated with rare cyclin-dependent kinase-like 5 (CDKL5) deficiency disorder (CDD) in patients of two years of age and older.
- February 2022 – Adalvo partnered with SK Pharma to create Arphio. It is a new business entity dedicated solely to orphan and rare disease medicines.
- August 2021 – The U.S. FDA accepted a new drug application from Agios Pharmaceuticals, Inc. for mitapivat for the treatment of adults with Pyruvate Kinase (PK) deficiency.
- May 2021 – The U.S. FDA granted a priority review to BCMA CAR-T cell therapy, an investigational therapy by Legend Biotech Corporation to treat relapsed or refractory multiple myeloma.
REPORT COVERAGE
The global research report provides a detailed analysis of the market and focuses on key aspects such as prevalence of major rare diseases in key countries, pipeline review of novel drugs, technological developments, and healthcare spending by key countries in 2019. Besides this, the report offers insights into the latest market trends and highlights key industry developments. In addition to the aforementioned factors, the report encompasses several factors that have contributed to the growth of the market in recent years.
Request for Customization to gain extensive market insights.
Report Scope & Segmentation
|
ATTRIBUTE |
DETAILS |
|
Study Period |
2021-2034 |
|
Base Year |
2025 |
|
Estimated Year |
2026 |
|
Forecast Period |
2026-2034 |
|
Historical Period |
2021-2024 |
|
Growth Rate |
CAGR of 12.57% from 2026 to 2034 |
|
Unit |
Value (USD billion) |
|
Segmentation |
By Therapy Type
|
|
By Drug Type
|
|
|
By Distribution Channel
|
|
|
By Region
|
Frequently Asked Questions
Fortune Business Insights says that the global market size was USD 210.26 billion in 2025 and is projected to reach USD 603.89 billion by 2034.
In 2025, the North America market size stood at USD 113.64 billion.
Recording a CAGR of 12.57%, the market will exhibit steady growth during the forecast period of 2026-2034.
The oncology segment is expected to be the leading segment in this market during the forecast period.
Rising prevalence of rare diseases and increasing R&D of orphan drugs by market players are major factors driving the growth of the market.
Bristol-Myers Squibb Company and F. Hoffmann-La Roche Ltd. are major players of the global market.
North America dominated the global market with a share of 54.04% in 2025.
Launch of advanced product offerings by prominent market players, increasing R&D initiatives, and increased awareness of rare diseases are expected to drive the adoption of orphan drugs in the global market.
Get 20% Free Customization
Expand Regional and Country Coverage, Segments Analysis, Company Profiles, Competitive Benchmarking, and End-user Insights.
Related Reports
-
US +1 833 909 2966 ( Toll Free )
-
Get In Touch With Us