Lateral Flow Assays Market Size, Share & Industry Analysis, By Product (LFA Readers [Benchtop and Handheld] and Kits & Reagents), By Technique (Sandwich Assays, Competitive Assays, and Multiplex Detection Assays), By End User (Hospitals & Clinics, Diagnostic Laboratories, Homecare Settings, and Others), and Regional Forecast, 2026-2034
KEY MARKET INSIGHTS
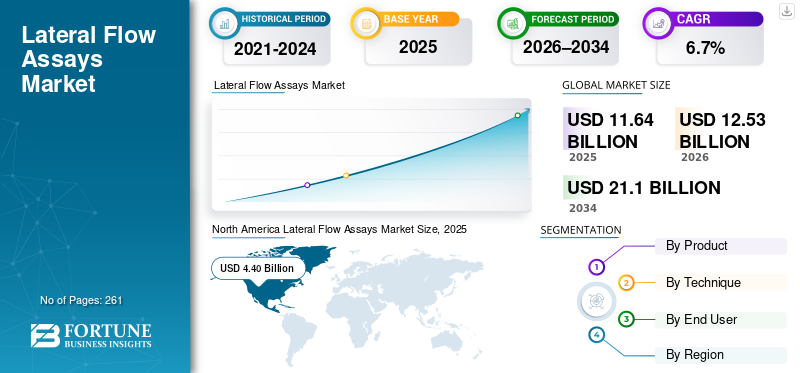
The global lateral flow assays market size was valued at USD 11.64 billion in 2025 and is projected to grow from USD 12.53 billion in 2026 to USD 21.10 billion by 2034, exhibiting a CAGR of 6.7% during the forecast period. North America dominated the lateral flow assays market with a market share of 37.8% in 2025.
Lateral flow assays are diagnostic devices that detect specific antigens or antibodies in liquid samples, including urine, saliva, and others. The growing prevalence of chronic conditions such as infectious disorders, rapid disease screening, and the development of healthcare infrastructure are resulting in a growing penetration rate of these products in the market. The growing aging population is further supporting the demand for diagnostic procedures among the patient population, thereby fueling the adoption rate of lateral flow assays in the market.
- For instance, according to the 2024 data published by the World Health Organization (WHO), an estimated 10.7 million people suffered from tuberculosis globally.
Furthermore, the rising integration of technological advancements in these products among the major players, such as Abbott, Thermo Fisher Scientific, Inc., among others, is further contributing to the demand for these devices in the market.
Download Free sample to learn more about this report.
Lateral Flow Assays Market Trends
Technological Advancements to Improve Result Accuracy and Data Management to be a Significant Market Trend
A prominent trend shaping the market is the incorporation of digital workflow and smartphone-based analysis platforms to improve result accuracy and data management. Conventional visually interpreted lateral flow assays are increasingly being complemented by portable readers that minimize subjectivity and enable quantitative or semi-quantitative analysis.
Furthermore, the players are introducing digital immunoassay products that pair lateral flow cartridges with small analyzers, enhancing sensitivity and clinical reliability. There is an increasing demand for smartphone-enabled devices, particularly for home-based testing and remote patient monitoring, further leading the focus of key players toward R&D activities to launch novel products in the market.
- In March 2025, Boots UK launched finger-prick Lateral Flow Tests (LFTs) to detect levels of vitamin D, iron, or cholesterol in people’s blood, as well as an influenza test.
Download Free sample to learn more about this report.
Market Dynamics
Market Drivers
Growing Burden of Infectious and Chronic Diseases to Boost Market Growth
The growing burden of chronic and infectious disorders remains the prominent driver for the global market. Infectious disorders, including malaria, HIV, tuberculosis, and hepatitis, affect hundreds of millions of people every year, especially in low- and middle-income countries such as Brazil, India, and others.
- For instance, according to 2025 data published by the World Health Organization (WHO), about 282 million malaria cases were reported globally in 2024.
This, along with the growing incidence of lifestyle-related chronic diseases, such as diabetes and cardiovascular diseases, has also fueled the adoption of these products for routine check-ups. The lateral flow-based cardiac marker tests are being used in emergency departments for rapid triage of patients with suspected myocardial infarction. Therefore, all the above factors are expected to drive the adoption rate of rapid, point-of-care, and home-based diagnostic testing, thereby supporting the global market size.
Other Prominent Drivers
- Low-cost, easy-to-use assays suitable for resource-limited settings.
- Expansion of OTC self-test kits and pharmacy-based diagnostics.
Market Restraints
Limited Analytical Sensitivity Compared to Other Techniques to Hamper Market Growth
There is an increasing adoption of these products for the diagnosis of various chronic and infectious diseases. However, lateral flow-based products face inherent technical limitations that hamper wider clinical acceptance in distinct applications.
The laboratory-based techniques, including molecular diagnostics, lateral flow-based products generally offer reduced analytical sensitivity and specificity, especially in early-stage disease detection or low-viral-load scenarios. This limitation has increased concerns among healthcare professionals regarding false-negative results, particularly for critical conditions requiring confirmatory diagnosis.
- For instance, according to various public health evaluations conducted during the COVID-19 pandemic, rapid antigen lateral flow-based tests demonstrated sensitivity ranging from 50% to 80%, compared to RT-PCR, with performance dropping further when viral loads were low.
Market Opportunities
Expansion of Healthcare Facilities to Create Lucrative Opportunities
There is a rapid expansion of clinical systems in developing countries, including India, Mexico, and others. The increasing prevalence of infectious disorders, the development of hospital infrastructure, rising number of diagnostic procedures are subsequently boosting the adoption of lateral flow assays in healthcare settings.
The preference for specialty clinics and diagnostic laboratories has increased for diagnostic procedures owing to their benefits, such as lower procedural costs, shorter patient stays, faster turnover times, and minimized risk of hospital-acquired infections.
- According to 2025 data published by Definitive Healthcare, there are about 10,000 active ambulatory surgical centers in the U.S.
Market Challenges
Limited Healthcare Access in Developing Countries to Limit Market Growth
There is a growing demand for routine check-ups among the patient population. However, shortage of technologically advanced products, limited healthcare expenditure, coupled with an inadequate reimbursement framework, especially in emerging nations, are resulting in limited access to healthcare facilities among the patient population.
Additionally, a limited number of clinical facilities and limited professionals, among others, are some of the crucial factors, resulting in the delayed diagnostic procedures among the patient population, particularly in emerging nations, including India, Brazil, among others.
- For instance, according to 2023 data published by The World Bank Group (WBG), about 4.5 billion people lack full access to essential health services globally.
SEGMENTATION ANALYSIS
By Product
Increasing Product Launches Led to Kits & Reagents Segment Dominance
Based on product, the market is classified into LFA readers and kits & reagents. LFA readers are further classified into benchtop and handheld.
To know how our report can help streamline your business, Speak to Analyst
The kits & reagents segment held the largest revenue share in 2025. The growth is due to the growing prevalence of infectious disorders among patients, resulting in a rising number of diagnostic procedures globally. This, along with the rising focus of key companies on launching novel kits and reagents, is further anticipated to contribute to the global lateral flow assays market growth.
- For instance, in July 2020, JOYSBIO (Tianjin) Biotechnology Co., Ltd., launched a lateral flow rapid COVID-19 neutralizing antibody test kit, which semi-quantitatively detects the level of neutralizing antibody against SARS-CoV-2 in serum, plasma, and whole blood samples.
The LFA readers segment is expected to grow at a CAGR of 7.4% over the forecast period.
By Technique
Increasing Number of Diagnostic Procedures Led to Dominance of Sandwich Assays Segment
Based on technique, the market is segmented into sandwich assays, competitive assays, and multiplex detection assays.
The sandwich assays segment dominated the global market and held the share of 58.1% in 2025. The growth is due to the increasing number of diagnostic procedures resulting in rising focus of key players towards R&D activities to launch novel products, thereby supporting the adoption rate of these products and market expansion globally.
- For instance, in September 2020, Abbott launched the BinaxNOW COVID-19 Ag Card, a rapid antigen lateral flow test designed for point-of-care and mass screening use. The product utilizes a sandwich immunoassay format to detect SARS-CoV-2 nucleocapsid protein and delivers results in approximately 15 minutes, significantly expanding access to rapid testing outside traditional laboratories.
The segment of multiplex detection assays is set to flourish with a growth rate of 7.0% across the forecast period.
By End User
Increasing Number of Hospitals & Clinics Led to Segmental Dominance
Based on end user, the market is classified into hospitals & clinics, diagnostic laboratories, homecare settings, and others.
The hospitals & clinics segment dominated the market in 2025. The development of healthcare infrastructure, rising number of hospitals and clinics, increasing adoption of advanced products, among others, are some of the key factors supporting the growth of the segment in the market. Furthermore, the segment is set to hold a 38.4% share in 2026.
- For instance, according to 2025 data published by Statistisches Bundesamt, it was reported that there are about 1,874 hospitals in Germany.
In addition, diagnostic laboratories’ end users are projected to grow at a 6.0% CAGR during the forecast period.
Lateral Flow Assays Market Regional Outlook
Based on region, the market has been studied across North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa.
North America
North America Lateral Flow Assays Market Size, 2025 (USD Billion)
To get more information on the regional analysis of this market, Download Free sample
The North America market held the dominant share in 2024, valued at USD 4.11 billion, and also took the leading share in 2025 with USD 4.40 billion. The growing prevalence of chronic conditions, developed hospital infrastructure, strong OTC and point-of-care adoption, among others, are some of the factors supporting the growth of the segment in the market.
- For instance, according to 2025 statistics published by the American Hospital Association (AHA), it was reported that there are about 6,093 hospitals in the U.S.
U.S. Lateral Flow Assays Market
Based on North America’s strong contribution and the U.S. dominance within the region, the U.S. market can be analytically approximated at around USD 4.18 billion in 2026, accounting for roughly 33.3% of global sales.
Europe
Europe is projected to record a growth rate of 4.7% in the coming years, which is the third highest among all regions, and is set to reach a valuation of USD 3.20 billion by 2026. The established regulatory framework and healthcare spending are likely to support the market growth.
U.K. Lateral Flow Assays Market
The U.K. market is estimated at around USD 0.57 billion in 2026, representing roughly 4.6% of global revenues.
Germany Lateral Flow Assays Market
Germany’s market is projected to reach approximately USD 0.60 billion in 2026, equivalent to around 4.8% of global sales.
Asia Pacific
Asia Pacific is estimated to reach USD 3.52 billion in 2026 and secure the position of the second-largest region in the market. The rapid testing programs and expanding healthcare access are likely to support the growth of the market.
Japan Lateral Flow Assays Market
The Japanese market is estimated at around USD 0.58 billion in 2026, accounting for roughly 4.6% of global revenues. Japan has historically reported a relatively high prevalence of chronic conditions, with a large number of diagnostic procedures.
China Lateral Flow Assays Market
China’s market is projected to be one of the largest globally, with revenues estimated at around USD 1.23 billion in 2026, representing roughly 9.8% of global sales.
India Lateral Flow Assays Market
The Indian market size is estimated at around USD 0.58 billion in 2026, accounting for roughly 4.6% of global revenues.
Latin America and Middle East & Africa
The Latin America and the Middle East & Africa regions are expected to witness moderate growth in this market during the forecast period. The Latin America market is set to reach a valuation of USD 0.63 billion in 2026. The Middle East & Africa is also expected to grow owing to growing adoption of advanced lateral flow assays in the market. The growth is due to the growing penetration in emerging economies in these regions.
GCC Lateral Flow Assays Market
The GCC is set to reach a value of USD 0.17 billion in 2026.
South Africa Lateral Flow Assays Market
The South Africa market is projected to reach around USD 0.09 billion in 2026, representing roughly 0.7% of global revenues.
Competitive Landscape
Key Industry Players
Growing focus on R&D Activities by Major Players to Support their Dominance
A robust and diversified product portfolio, along with a significant focus on strategic initiatives globally, is one of the crucial factors supporting the dominance of these companies in the market. Abbott and Thermo Fisher Scientific, Inc., were major companies in the market in 2025. Moreover, the growing focus of key companies on R&D activities is likely to strengthen their presence, further supporting the global lateral flow assays market share.
- For instance, in September 2025, Abbott launched of Panbio COVID-19 Antigen Self-Test for the detection of the SARS-CoV-2 virus in adults and children with or without symptoms.
Other key players, including F. Hoffmann-La Roche Ltd., and others, are also growing in the market, primarily due to their increasing focus on acquisitions among other players to strengthen their presence in the market.
List of Key Lateral Flow Assays Companies Profiled in Report
- Abbott (U.S.)
- Thermo Fisher Scientific Inc. (U.S.)
- Hoffmann-La Roche Ltd. (Switzerland)
- Siemens Healthineers AG (Germany)
- QuidelOrtho Corporation (U.S.)
- BIOMÉRIEUX (France)
- JOYSBIO (Tianjin) Biotechnology Co., Ltd. (China)
- Milenia Biotec GmbH (Germany)
- Meizheng (China)
- Danaher Corporation (U.S.)
KEY INDUSTRY DEVELOPMENTS
- April 2024: QuidelOrtho Corporation received U.S. Food and Drug Administration (FDA) 510(k) clearance for its QuickVue COVID-19 test.
- August 2023: Anbio, a provider of innovative diagnostic solutions, launched the Anbio LFA solution, an innovative, easy-to-use diagnostic product. Anbio's LFA solution, or Lateral Flow Assay, is a diagnostic technique for detecting antigens (s) such as viral proteins and cancer biomarkers.
- July 2023: SEKISUI Diagnostics launched the OSOM COVID-19 Antigen Home Test to diagnose COVID-19 at home, to strengthen its global presence.
- July 2023: Hoffmann-La Roche Ltd., launched a combined SARS-CoV-2 and Influenza A/B rapid antigen test using lateral flow sandwich assay technology. The multiplex test was designed to support differential diagnosis of respiratory infections in point-of-care settings.
- October 2022: Roche launched its next-generation portfolio SARS-CoV-2 rapid antigen tests (“2.0”) for self-testing and professional use in countries accepting the CE Mark.
- September 2022: Abbott expanded its rapid diagnostics portfolio with the launch of the BinaxNOW COVID-19 Ag Self Test, optimized for at-home use. The product retained the sandwich immunoassay configuration while improving usability and packaging for consumer markets.
- August 2021: QuidelOrtho Corporation launched the QuickVue At-Home OTC COVID-19 Test, a visually read lateral flow antigen test employing sandwich immunoassay design. The product expanded Quidel’s presence in the consumer self-testing segment and supported the growing shift toward home-based diagnostics.
REPORT COVERAGE
The global lateral flow assays market report provides a detailed market analysis and focuses on key aspects such as leading companies and market segmentation, including product, technique, and end user. Besides this, the global report offers insights into the market growth trends and highlights key industry developments. In addition to the aforementioned factors, the report encompasses several factors that have contributed to the growth and advancement of the market over recent years.
Request for Customization to gain extensive market insights.
Report Scope & Segmentation
| ATTRIBUTE | DETAILS |
| Study Period | 2021-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2021-2024 |
| Growth Rate | CAGR of 6.7% from 2026-2034 |
| Unit | Value (USD Billion) |
| Segmentation | By Product, Technique, End User, and Region |
| By Product |
|
| By Technique |
|
| By End User |
|
| By Region |
|
Frequently Asked Questions
Fortune Business Insights says that the global market size was USD 11.64 billion in 2025 and is projected to reach USD 21.10 billion by 2034.
In 2025, the North America regional market value stood at USD 4.40 billion.
Growing at a CAGR of 6.7%, the market will exhibit steady growth over the forecast period (2026-2034).
By product, the kits & reagents is the leading segment in this market.
The introduction of novel lateral flow assays is one of the major factors driving the markets growth.
Abbott and Thermo Fisher Scientific Inc., are the major players in the global market.
North America held the highest market share in 2025.
The growing prevalence of chronic diseases, the growing number of diagnostic procedures, among others, are some of the prominent factors expected to boost the adoption of these products globally.
Get 20% Free Customization
Expand Regional and Country Coverage, Segments Analysis, Company Profiles, Competitive Benchmarking, and End-user Insights.
Related Reports
-
US +1 833 909 2966 ( Toll Free )
-
Get In Touch With Us