Smart Manufacturing in Pharma Market Size, Share & Industry Analysis, By Component (Solution {Manufacturing Execution & Operations Management, Quality Digitalization and Others}, and Services), By Deployment (Cloud-Based, On Premise and Hybrid), By Technology (Automation, IIoT & Connectivity, Digital Twins & Simulation, AI/ML & Advanced Analytics, & Others), By Application (Production Management, Batch Record Management, Weighing & Dispensing Material Management, & Others), By End User (Pharmaceutical & Biotechnology Companies, & Others) and Regional Forecast, 2026-2034
KEY MARKET INSIGHTS
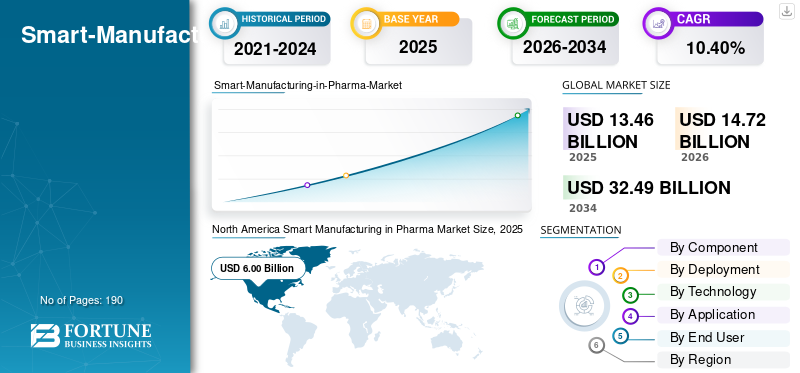
The smart manufacturing in pharma market size was valued at USD 13.46 billion in 2025. The market is projected to grow from USD 14.72 billion in 2026 to USD 32.49 billion by 2034, exhibiting a CAGR of 10.40% during the forecast period. North America dominated the global smart manufacturing in pharma market with a share of 44.57% in 2025.
The market is anticipated to experience strong growth in the coming years. The pharmaceutical sector faces urgent challenges driven by evolving patient demographics, diverse product types, regulatory scrutiny, and performance pressure. These factors collectively drive demand for growth-efficient pharmaceutical production processes to meet production targets and enable continuous improvement through digital and automation solutions. As product portfolios become more complex, manufacturers are investing in connected factories that can standardize execution, automate quality controls and enable real-time visibility across equipment, processes, and sites. Furthermore, innovative solutions that incorporate new technologies in smart manufacturing in pharma, optimizing manufacturing workflows also drive market growth.
- For instance, in May 2025, Rockwell Automation announced the release of Factory Talk PharmaSuite 12.00, positioning it to help pharmaceutical and biopharmaceutical manufacturers simplify system management and improve flexibility, which reflects rising demand for scalable, secure manufacturing execution capabilities in pharma plants. Such developments are anticipated to boost the overall market growth.
Furthermore, leading players in the smart manufacturing in the pharma industry, such as Siemens, Körber AG and Teva Pharmaceutical Industries Ltd., are directing their resources toward technological advancement and new product launches to strengthen their market positions.
Download Free sample to learn more about this report.
SMART MANUFACTURING IN PHARMA MARKET TRENDS
Serialization & Traceability Mandates as a Core Application is Prominent Market Trend
A significant trend observed in the market is an increasing focus on serialization & traceability mandates. Regulators are increasingly requiring unit-level identification and electronic, interoperable trace data across the drug supply chain, which forces manufacturers and trading partners to upgrade packaging lines, integrate EPCIS/event data exchange, and implement exception-handling workflows to avoid distribution disruptions. As these rules tighten and timelines advance, companies move beyond “basic serialization” toward end-to-end traceability, which raises demand for advanced smart manufacturing and traceability platforms that can connect shop-floor packaging operations with downstream distribution systems.
- For instance, under the 2024 DSCSA Mandate, pharmaceutical companies must assign a unique identifier to each saleable unit of a drug. This identifier should include vital information such as the product's lot number, expiration date and a unique serial number for each unit. Such developments are expected to boost market growth.
Download Free sample to learn more about this report.
MARKET DYNAMICS
MARKET DRIVERS
Increasing Regulatory Compliance and Data-Integrity Requirements Are Driving Market Growth
The growing trend of digitalization and increasing regulatory compliance are primarily driving smart manufacturing in pharma market growth. Manufacturers are required to maintain accurate, traceable electronic records throughout the entire production lifecycle. As scrutiny on deviations, batch documentation and data governance increases, companies are adopting smart manufacturing tools such as connected MES, electronic batch records, automated quality workflows and real-time monitoring to reduce manual errors, improve traceability and speed up investigation and release cycles. These factors result in greater pharmaceutical output to cater to market demand. Key companies are focusing on operational efficiency in their manufacturing processes, using these smart technologies to maximize output.
- For instance, in July 2025, the Association for Accessible Medicines declared that Amphastar Pharmaceuticals, Inc. invested in domestic manufacturing, intending to quadruple its production in the next three to five years. Such factors highlight the expected drivers of market growth.
MARKET RESTRAINTS
Cyber Security Threats and Ransomware Disruption to Hamper Market Growth
Smart manufacturing adoption in pharmaceutical companies is restrained mainly as manufacturing plants become more connected, OT/IT cybersecurity exposure increases, and the fear of ransomware or operational disruption pushes manufacturers to add security-by-design controls and segmentation before scaling. These factors raise implementation cost and extend timelines. Additionally, organizational readiness becomes a bottleneck as Pharma 4.0 programs are built on data-integrity foundations that require cross-functional governance, new operating models, and capability development. This results in many companies struggling to move from pilots to enterprise scale at speed.
- For instance, in June 2025, Honeywell International Inc. reported in its Cybersecurity Threat Report that ransomware attacks jumped by 46% from Q4 2024 to Q1 2025. Malware and ransomware increased significantly, including a 3,000% spike in the use of one Trojan designed to steal credentials from industrial operators. Such factors discourage pharmaceutical companies from adopting smart manufacturing technologies and limit their growth potential.
MARKET OPPORTUNITIES
Integration of Innovative Technologies to Improve Yield and Unlock New Growth Opportunities
The growing integration of innovative technologies, such as AI-enabled digital twins for predictive monitoring and anomaly detection, offers significant growth opportunities for the market and improved uptime in regulated plants. As processes become more complex and manufacturers aim to reduce deviations and batch losses, there is a growing opportunity for digital twins to combine real-time plant data with AI to detect anomalies early and support data-driven operational decisions. This is especially attractive in pharma as better fault detection and predictive maintenance can directly improve reliability and consistency, key drivers of compliance and supply continuity, so solutions that operationalize digital twins in validated environments are probable to see rising demand.
- For instance, in January 2026, WuXi Biologics launched PatroLab, a digital twin platform designed to support bioprocess development and manufacturing. The system integrated real-time process monitoring, Raman-based Process Analytical Technology (PAT), and predictive in silico modelling to enable smarter analytics, proactive control and faster decision-making across biologics programs. Such developments offer market growth opportunities.
MARKET CHALLENGES
High Upfront Capital Expenditure Poses a Significant Challenge to Market Growth
One of the major challenges in the market is the higher upfront implementation cost. A smart pharma factory typically requires large, bundled investments, particularly in automation and OT upgrades, sensor/PAT infrastructure, secure networks, data platforms and validation and change-control work. This results in a long and uncertain payback period. As capex requirements rise, manufacturers often delay plant modernization, scale deployments more slowly, or limit upgrades to a few critical lines/sites, which directly slows broad adoption of smart manufacturing solutions across the industry.
- For instance, in April 2025, GMP Pros reported that, for a typical mid-sized pharmaceutical facility, MES implementation costs range from USD 1.5 to 3 million, such high implementation costs are slowing adoption.
Segmentation Analysis
By Component
New Product Launches in Solution Segment to Position Them in Leading Position
Based on the component, the market is categorized into solutions and services.
Among these, the solution segment accounted for the largest smart manufacturing in pharma market share in pharma in 2025.The segment dominated, as pharmaceutical companies typically buy smart manufacturing as an integrated system of record for MES/EBR/QMS connectivity, data capture, workflows and lifecycle tools. This reduces manual interventions, improves traceability and shortens validation cycles across regulated operations. Such bundled solutions, as in implementation/support, drives growth in the segment. Additionally, new product launches by key companies to digitalize manufacturing workflows, driving growth in the segment.
- For instance, in June 2025, Rockwell Automation, Inc. announced that Swiss company Sintetica SA adopted the FactoryTalk PharmaSuite Manufacturing Execution System (MES) from Rockwell Automation as a driving force behind its expansive digital transformation. Such developments are expected to drive the segment's growth.
The services segment is expected to grow at a CAGR of 8.37% over the forecast period.
To know how our report can help streamline your business, Speak to Analyst
By Deployment
Innovative Product Launches in Cloud-Based Platforms to Drive Segmental Growth
Based on deployment, the market is segmented into cloud-based, on-premises and hybrid.
Among these, cloud-based deployment accounted for the largest share. Cloud-based deployment dominated as manufacturers demand for faster rollouts and easier multi-site standardization. These factors have resulted in a shift toward cloud-based models that improve scalability and reduce the operational burden of maintaining complex manufacturing applications.
- For instance, in April 2025, Honeywell launched TrackWise Manufacturing, an Artificial Intelligence (AI)-assisted, cloud-native platform designed to transform how life sciences companies manage, automate and digitalize operations.
In addition, the hybrid medicine market is projected to grow at a CAGR of 8.92% over the study period.
By Technology
Reduced Eros due to Automation Reinforces Segmental Growth
Based on the technology, the market is segmented into automation, IIoT & connectivity, digital twins & simulation, AI/ML & advanced analytics, robotics & autonomous systems, and others.
In 2025, automation segment dominated the market in terms of technology. Automation dominates as pharma plants prioritize repeatability and controlled execution to reduce deviations and maintain GMP compliance. This encourages pharmaceutical companies to invest in automation platforms that connect equipment, enforce process control and enable reliable data capture, since automated control loops and standardized execution directly improve throughput, consistency in quality, and audit readiness compared with manual interventions.
- For instance, in May 2025, Emerson, a technology leader delivering advanced automation solutions, launched DeltaV, an automation software, to assist manufacturers. The application makes it easier for organizations to select and connect critical tools necessary to deliver quality treatments to patients faster and safely.
AI/ML & advanced analytics is projected to grow at a CAGR of 16.02% during the forecast period.
By Application
Varied Application of Smart Manufacturing in Production Management to Fuel Segmental Growth
Based on the application, the market is segmented into production management, batch record management, weighing & dispensing material management, digital quality management, laboratory informatics (QC/QA) and others.
In 2025, production management dominated the market based on application. The segment's share is attributed to its critical role in operations. It schedules, allocates resources, executes and assesses performance across lines and sites. Companies first invest in production management capabilities to improve visibility, standardize execution and reduce disruption, which then serves as the foundation for batch records, quality workflows and analytics. Highlighting the critical nature of such applications, many key companies are participating in strategic collaborations to strengthen their market position.
- For instance, in October 2025, Siemens and Capgemini expand their strategic partnership to co-develop AI-native digital solutions for product engineering, manufacturing, and operations. The partnership aimed to deliver industry-specific solutions, automation, electrification, and a sustainability portfolio with Capgemini’s engineering capabilities, industry knowledge, and business transformation expertise.
Laboratory Informatics (QC/QA) is projected to grow at a CAGR of 12.14% during the forecast period.
By End User
Bulk Utilization by Pharmaceutical & Biotechnology Companies Leads to Segment’s Growth
Based on end user, the market is segmented into pharmaceutical & biotechnology companies, CDMOs & CMOs, and others.
Pharmaceutical and biotechnology companies segment dominated the market. Higher adoption by these operating entities, as they own the largest regulated manufacturing footprints, was a critical factor in their high market share. They bear the highest direct risk from batch failures, compliance findings, and supply disruptions. As a result, they invest more aggressively in smart manufacturing to protect the brand, ensure supply continuity, and scale new capacity.
- For instance, in December 2025, Eli Lilly invested more than USD 6.00 billion in a next-generation API manufacturing facility in the U.S. Such developments reflect how large pharma drives the bulk of modernization and smart-factory spending.
The CDMOs & CMOs segment is projected to grow at a CAGR of 12.65% over the study period.
Smart Manufacturing in Pharma Market Regional Outlook
By geography, the market is categorized into Europe, North America, Asia Pacific, Latin America, and the Middle East & Africa.
North America
North America Smart Manufacturing in Pharma Market Size, 2025 (USD Billion)
To get more information on the regional analysis of this market, Download Free sample
North America held the dominant share in 2024, valued at USD 5.49 billion, and maintained its leading position in 2025, with a value of USD 6.00 billion. The market in North America is expected to grow significantly over the forecast period, driven by improvements in quality and the avoidance of compliance issues. Supply chain resilience after shortages is pushing more automation and real-time monitoring. Strong budgets and faster adoption of AI/MES also accelerates the spending.
U.S. Smart Manufacturing in Pharma Market
Given the North America’s substantial contribution, the U.S. market can be estimated at around USD 6.02 billion in 2026, accounting for roughly 40.91% of the global market.
Europe
Europe is projected to grow at 9.90% over the coming years, the second-highest among all regions, and reach a valuation of USD 3.76 billion by 2026. The area is expected to experience robust growth driven by the strong need for stronger data integrity across manufacturing and quality.
U.K. Smart Manufacturing in Pharma market
The U.K. market in 2026 is estimated at around USD 0.58 billion, representing roughly 3.96% of the global market.
Germany Smart Manufacturing in Pharma market
Germany’s market is projected to reach approximately USD 0.97 billion in 2026, equivalent to around 6.61% of the global market.
Asia Pacific
Asia Pacific is estimated to reach USD 3.57 billion in 2026 and secure the position of the third-largest region in the market. The region's growth is driven by higher production volumes, which are putting pressure to automate and reduce batch failures.
Japan Smart Manufacturing in Pharma Market
The Japanese market in 2026 is estimated at around USD 0.87 billion, accounting for approximately 5.88% of the global market.
China Smart Manufacturing in Pharma Market
China’s market is projected to be among the largest worldwide, with 2026 revenues estimated at around USD 1.20 billion, representing approximately 8.15% of global sales.
India Smart Manufacturing in Pharma Market
The Indian market is estimated at around USD 0.29 billion in 2026, accounting for roughly 1.99% of global revenue.
Latin America and the Middle East & Africa
The Latin America and Middle East & Africa regions are expected to witness moderate growth in this market space during the forecast period. The Latin America market is set to reach a valuation of USD 0.44 billion in 2026. The region is experiencing market growth driven by increased government support. In the Middle East & Africa, the GCC is set to reach USD 0.19 billion in 2026.
South Africa Smart Manufacturing in Pharma market
The South African market is projected to reach approximately USD 0.06 billion by 2026, accounting for roughly 0.40% of global revenue.
COMPETITIVE LANDSCAPE
Key Industry Players
Strategic Acquisitions by Key Players to Propel Market Progress
The global smart manufacturing in pharma market is highly consolidated, with companies such as Siemens, Körber AG, Teva Pharmaceutical Industries Ltd, and Honeywell holding a considerable market share. Strategic partnerships, new product launches, technological advancements, and increased investments and strategic acquisitions in the sector drive these companies' market share gains.
- For instance, in June 2025, Honeywell acquired Sundyne for USD 2.16 billion. Such developments are aimed at driving market growth.
Other notable players in the global market include Emerson, AVEVA, and ABB These companies are expected to prioritize technological advancements, strategic collaborations, and new product launches to strengthen their position during the forecast period.
LIST OF KEY SMART MANUFACTURING IN PHARMA COMPANIES PROFILED
- Körber AG (Germany)
- Rockwell Automation. (U.S.)
- Siemens. (Germany)
- Emerson (U.S.)
- AVEVA (U.K.)
- Honeywell (U.S.)
- ABB (Switzerland)
- Amazon Web Services. (U.S.)
- Dassault Systèmes (France)
- Aspen Technology (U.S.)
KEY INDUSTRY DEVELOPMENTS
- December 2025: Intelligent Bio Solutions Inc. partnered with Syrma Johari MedTech Ltd. to support and scale the production of its Intelligent Fingerprinting Drug Screening Reader. The collaboration is expected to support long-term margin improvement.
- November 2025: Teva Pharmaceutical Industries Ltd. Launched Teva Rise, a global open innovation platform designed to harness the power of a variety of disruptive technologies including AI, industry 4.0 smart manufacturing, digital health and biotech, by connecting startups and technology companies with Teva’s business units. The initiative aimed to drive patient impact and business transformation.
- August 2025: Körber launched the latest version of its PAS-X Manufacturing Execution System. The new PAS-X MES 3.4 stands out with its web-based, scalable platform, intuitive user experience, AI support, and smart tools for deployment, monitoring, and long-term lifecycle management.
- February 2025: Capula, launched its Smart Digital Operations (SDO) service. The approach integrated intelligent technologies and digital solutions to digitalize, optimize and streamline industrial operational processes in pharmaceutical manufacturing. The development addressed six key industry challenges, disruption management, process efficiency, net zero, product compliance, asset health and performance, and supply chain agility.
REPORT COVERAGE
The global smart manufacturing in pharma market analysis includes a comprehensive study of market size & forecast across all market segments covered in the report. It contains details on the market dynamics and trends expected to drive the global smart manufacturing in pharma market over the forecast period. It provides information on key aspects, including technological advancements and new product launches. Additionally, it details partnerships, mergers & acquisitions, and key industry developments. The global market research report also provides a detailed competitive landscape, including market share and profiles of major operating players.
Request for Customization to gain extensive market insights.
Report Scope & Segmentation
| ATTRIBUTE | DETAILS |
| Study Period | 2021-2034 |
| Base Year | 2025 |
| Forecast Period | 2026-2034 |
| Historical Period | 2021-2024 |
| Growth Rate | CAGR of 10.40% from 2026 to 2034 |
| Unit | Value (USD Billion) |
| Segmentation | By Component, Deployment, Technology, Application, End User, and Region |
| By Component |
|
| By Deployment |
|
| ByTechnology |
|
| By Application |
|
| By End User |
|
| By Region |
|
Frequently Asked Questions
According to Fortune Business Insights, the global market value stood at USD 13.46 billion in 2025 and is projected to reach USD 32.49 billion by 2034.
In 2025, the North America market value stood at USD 6.00 billion.
The market is expected to grow at a CAGR of 10.40% over the forecast period of 2026-2034.
The solutions segment is expected to lead the market, by component.
Increasing focus on reducing errors in medication management is driving market growth.
Siemens, Körber AG, Teva Pharmaceutical Industries Ltd, and Honeywell are the major market players in the global market.
North America dominated the market in 2025.
Get 20% Free Customization
Expand Regional and Country Coverage, Segments Analysis, Company Profiles, Competitive Benchmarking, and End-user Insights.
-
US +1 833 909 2966 ( Toll Free )
-
Get In Touch With Us