Viral Vaccines Market Size, Share & Industry Analysis, By Form (Liquid and Lyophilized), By Container Format (Vials and Prefilled Syringes), By Biosafety Level Type (Biosafety Level 1, Biosafety Level 2, Biosafety Level 3, and Biosafety Level 4), and Regional Forecast, 2026-2034
Viral Vaccines Market Size and Industry Overview
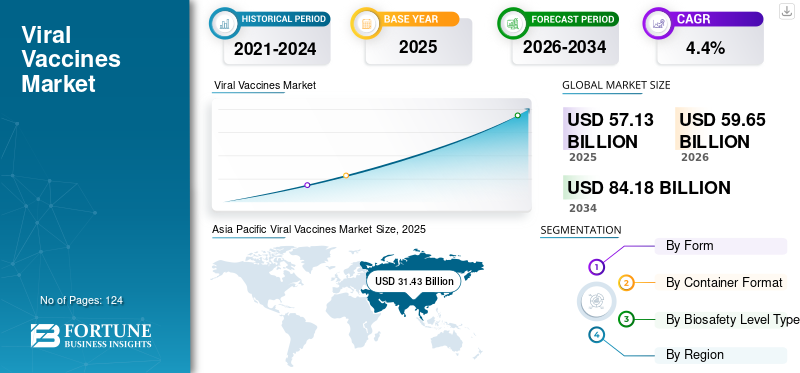
The global viral vaccines market size of USD 57.13 billion in 2025 and is projected to expand from USD 59.65 billion in 2026 to USD 84.18 billion by 2034, growing at a CAGR of 4.40% over the forecast period. Asia Pacific dominated the viral vaccines market with a market share of 55.01% in 2025. Moreover, the U.S. viral vaccines market size is projected to grow significantly, reaching an estimated value of USD 26.14 billion by 2030, driven by growing number of initiatives to rollout vaccination plans for the prevention of viral diseases.
The incidence of these viral diseases varies from region to region as well as from season to season, which often causes fluctuations in the demand for these products across the globe. For instance, as per an article published by Physiopedia in 2022, Japanese encephalitis is the most common cause of encephalitis in Asia due to favorable conditions offered by this region for the growth of this virus. Some other viruses that need geographical and seasonal variation for growth include dengue virus, Enterovirus (EV71), rabies, Nipah virus, herpes simplex, West Nile virus, mumps, and many more.
Over decades, the range and availability of viral vaccines have been expanding globally, with the development of new products against viral diseases, such as hepatitis E, dengue fever, Ebola, and many more, that primarily affect lower-income countries. Organizations, such as the Coalition for Epidemic Preparedness Innovations (CEPI), Serum Institute of India, and many more are leveraging innovative platforms by supporting research projects to develop products against emerging diseases. These factors are anticipated to drive the growth of the viral vaccines market during the forecast period.
Download Free sample to learn more about this report.
Global Viral Vaccines Market Snapshot & Highlights
Market Size & Forecast:
- 2025 Market Size: USD 57.13 billion
- 2026 Market Size: USD 59.65 billion
- 2034 Forecast Market Size: USD 84.18 billion
- CAGR: 4.4% from 2026–2034
Market Share:
- Asia Pacific dominated the viral vaccines market with a 55.01% share in 2025, driven by the rising prevalence of viral diseases such as hepatitis, HIV, HPV, along with strong government-led immunization initiatives across countries like Japan, China, and India.
- By container format, the vials segment is expected to retain its largest market share owing to its cost-effectiveness over prefilled syringes and continued preference by manufacturers for large-scale immunization programs, despite the rising trend of prefilled syringes for patient safety and dose accuracy.
Key Country Highlights:
- United States: Increasing initiatives to roll out comprehensive vaccination plans against viral diseases and strong R&D investment in next-generation vaccine technologies.
- Europe: Rising government expenditure and public acceptance of immunization programs, including in vaccine-hesitant countries like France, driving demand for viral vaccines.
- China: Expanding vaccination coverage for diseases such as hepatitis, HPV, supported by large-scale domestic manufacturing capabilities and government funding.
- Japan: Strategic government partnerships to procure vaccines for regional immunization campaigns, including oral polio vaccines and innovative research collaborations for emerging viral threats.
Viral Vaccines Market Trends
Gradual Shift from Vials to Prefilled Syringes to Determine Market Growth
One of the most prevailing trends witnessed in this market is the inclination of market players to manufacture these products in Prefilled Syringe (PFS) form rather than in vial form owing to the benefits the former offers. Compared to vials, PFS offers various advantages in terms of speed, disposal, wastage, and patient safety as it contains premeasured and accurate doses that reduce dosing errors and risk of microbial contamination. These factors have continued to gain favor among vaccine manufacturers and healthcare providers for creating Prefilled Syringes (PFS).
- In July 2022, Novavax entered an agreement with SK Bioscience to develop and commercialize a version of the COVID-19 vaccine containing the Omicron variant in prefilled syringes.
This gradual shift of manufacturers from vials to prefilled syringes due to the benefits the latter provides to the healthcare provider in terms of accuracy in delivering vaccines to patients is expected to favor the market growth during the forecast period.
Download Free sample to learn more about this report.
Viral Vaccines Market Growth Factors
Increasing Incidence of Viral Infections to Propel Market Growth
A key market driver is the increasing incidence of viral infections globally. These infections are caused by viruses whose frequency and distribution vary from region to region. A few notable examples that have gathered the attention of the public health community and the population at large include COVID-19, Ebola, SARS, influenza, Zika, yellow fever, Human Immunodeficiency Virus (HIV/AIDS), Human Papillomavirus (HPV), viral gastroenteritis, varicella, and viral hepatitis.
- According to the European Centre for Disease Prevention and Control 2023 data, 3 million new cases of hepatitis B and C are registered per year globally, while in the EU/EEA there are approximately 6 million people living with chronic hepatitis B and C infections.
Some of the chronic viral infections create a severe public health problem, as they significantly increase the risk of chronic diseases, such as cancer.
- As per a report published by the HPV Information Center in 2021, HPV infection is a well-established cause of cervical cancer, and HPV types 16 and 18 are responsible for about 70% of all cervical cancer cases worldwide. The annual number of cervical cancer cases caused by HPV in India was 123,907 in 2021, and this number varies from country to country. An increase in the incidence of these viral infections will boost the demand for viral vaccines as a preventive measure, driving the market growth during the forecast period.
Surge in Initiatives by Government Authorities for Immunization to Drive Market Growth
Globally, government authorities are actively focused on the introduction of new programs to eradicate and prevent certain viral diseases. The promotion of vaccination drives is one of the key effective measures to combat these viral diseases.
- In April 2022, the Global Polio Eradication Initiative (GPEI) raised the urgency to renew eradication strategy for polio at a virtual event. The new strategy, if fully funded, will witness the vaccination of 370 million children annually for the next five years and the continuation of global surveillance activities for polio and other diseases in 50 countries. Such initiatives by various regulatory bodies will lead to the adoption of early preventive measures and eventually eliminate highly infectious diseases.
RESTRAINING FACTORS
Stringent Pricing Control to Hinder Market Growth
Despite the increasing demand for viral vaccines in the global market, the stringent regulatory control over the prices of these products is a substantial restraining factor for the market’s growth. This may prevent many established biopharmaceutical companies from investing in this market to avoid financial loss.
- For instance, in Japan, the pharmaceutical industry is subject to government-mandated annual price reductions of pharmaceutical products, including certain vaccines. Furthermore, the government can order re-pricings for specific products defined under applicable re-pricing rules if it determines that the use of such products will exceed certain thresholds. Such restrictions from government bodies may negatively influence market players' interest to invest further in this market to prevent business loss, thereby restricting the market’s growth.
Moreover, several pharmaceutical companies have introduced vaccines against Coronavirus during the pandemic, out of which only a few were approved, which led to a significant difference in pricing across various regions.
- According to an article published by Reuters in October 2022, Pfizer expressed plans to roughly quadruple the price of its COVID-19 vaccine to about USD 110 – USD 130 per dose after the U.S. government’s current purchase programs expire. These pricing pressures and market access challenges continue to vary, which may slow down the growth of the global market during the forecast period.
Viral Vaccines Market Segmentation Analysis
By Form Analysis
Liquid Vaccines to Gain Traction Due to Low Manufacturing Cost
On the basis of form, the market is divided into liquid and lyophilized.
The liquid segment dominated the global market in 2022 due to various benefits, such as the requirement for lesser processing and handling time. Moreover, as it incurs minimal manufacturing cost, many viral vaccines are commercialized in liquid form. According to an article published by the MDPI (Pharmaceuticals) in June 2023, it was concluded that even though the cold chain requirements of the lyophilized formulations were significantly lower than those of the liquid formulations, the process requirements of the latter were relatively lower than those of the lyophilized one, which makes liquid formulations more suitable for early clinical use.
The lyophilized segment is estimated to record a robust CAGR due to its longer shelf life and better stability than liquid form. As per an article published by the European Pharmaceutical Review in January 2022, U.S.-based scientists from the University of Massachusetts Lowell (UMass Lowell) were granted USD 930,000 to develop a freeze-drying technique for mRNA-based COVID-19 vaccines. These products can be transported and stored at room temperature. The benefits provided by lyophilized form of vaccines are expected to drive the segment’s growth during the forecast period.
To know how our report can help streamline your business, Speak to Analyst
By Container Format Analysis
Cost-Effectiveness of Conventional Vials to Propel Their Use
Based on container format, the market is bifurcated into vials and prefilled syringes.
The vials segment will hold the maximum global viral vaccines market share throughout the forecast period due to its cost-effectiveness over prefilled syringes. This has significantly influenced the focus of market players in terms of the manufacturing and commercialization of these products in vial form. These factors will contribute to the segment’s growth in the global market. For instance, in June 2022, GSK plc received the U.S. FDA approval for Priorix (Measles Mumps and Rubella Vaccine, Live) for 12 months and older individuals. In the market, the vaccine is commercialized in the vial format with a subcutaneous route of administration. Such new product launches in the market will propel the segment’s growth.
The Prefilled Syringes (PFS) segment will register the highest CAGR during the forecast period as it offers multiple advantages over the conventional vial format, such as simplified vaccine administration and reduction in dosage errors. It also minimizes the potential for microbial cross-contamination and pathogen transmission, which can happen when multiple doses are withdrawn from a single vial.
By Biosafety Level Type Analysis
Introduction of COVID-19 Vaccine to Boost Launch of Biosafety Level Type 3 Vaccines
By biosafety level type, the market is classified into biosafety level 1, biosafety level 2, biosafety level 3, and biosafety level 4.
The biosafety level 3 segment generated the highest revenue in 2022, and the growth is majorly driven by the surge in COVID-19 vaccinations across the globe from 2020 onwards. According to an article published by the CATO INSTITUTE in January 2023, roughly 655 million doses of COVID-19 vaccines were rolled out, which were enough for covering 80% of the population. Through 2022, COVID-19 vaccination efforts in the U.S. prevented more than 18 million hospitalizations and 3 million additional deaths. Such huge utilization of COVID-19 vaccines strongly contributed to the segment’s growth.
The biosafety level 2 segment is anticipated to record a significant CAGR during the forecast period. The segment includes products that offer protection against viral diseases, such as chickenpox, measles, mumps, rotavirus, hepatitis, influenza, and many more. The rising incidence of these diseases will increase the demand for these vaccines during the forecast period.
Biosafety level 1 and biosafety level 4 segments accounted for a comparatively smaller market share in 2022 and are estimated to grow steadily during the forecast period. This is owing to increased focus of key players on introducing novel products by accelerating the research and development of their pipeline candidates.
REGIONAL ANALYSIS
In terms of region, the global market is studied across North America, Europe, Asia Pacific, and the rest of the world.
Asia Pacific
Asia Pacific Viral Vaccines Market Size, 2025 (USD Billion)
To get more information on the regional analysis of this market, Download Free sample
Asia Pacific dominated the market with a valuation of USD 31.43 billion in 2025 and is projected to reached USD 32.81 billion in 2026. and is likely to remain dominant throughout the forecast period. The rising prevalence of major viral diseases, such as hepatitis, HIV, HPV, and many more is a key factor for market’s growth. In addition, the growing support of government bodies through the introduction of vaccination drives to control the incidence rate and eradicate the disease eventually is strongly contributing to the market’s growth across the region. For instance, in December 2021, the government of Japan announced a USD 4.3 million grant for the procurement of essential oral polio vaccines as part of its continued support to Pakistan. The latest grant will be utilized to procure 24 million oral doses of these products.
North America
North America held the second-largest share of the market in 2022. The robust usage of viral vaccines and growing number of initiatives to roll out vaccination plans for viral diseases are expected to drive the market’s growth in this region. For instance, in October 2022, Ottawa Public Health launched an annual influenza vaccination campaign for the general public.
Europe
Europe held a significant market share owing to the rising R&D initiatives for these vaccines and growing government expenditure for preventing these infectious diseases. According to an article published by the National Institutes of Health (NIH) in 2022, it was observed that the acceptance of vaccination programs against the COVID-19 infection was increasing. Furthermore, it also states that France, which is amongst the world’s most vaccine-hesitant countries, were opting for COVID-19 vaccines. The rising adoption rate among the general population will augment the regional market’s growth.
The rest of the world
The rest of the world is estimated to record a comparatively lower CAGR, including Latin America, the Middle East, and Africa. Some factors lowering the adoption rate of viral vaccines are the limited supply and lack of manufacturers in low-income countries, such as Iran, Iraq, and Kenya. According to an article published by the United Nations International Children's Emergency Fund (UNICEF), Iraq received 336,000 doses of AstraZeneca COVID-19 vaccine in March 2021 through the COVAX facility.
List of Key Viral Vaccines Market Companies
Strong COVID-19 Vaccine Sales by Pfizer, Inc. to Pivot Company to Peak Position
In terms of the competitive landscape, the market is consolidated, with a few market players holding a majority share of the market. Pfizer, Inc. has a dominant market position owing to the strong sales of its COVID-19 vaccine, Comirnaty, which received regulatory approvals from the U.S. FDA in August 2021.
Merck & Co. Inc. is one of the major players in the market due to its diversified range of products to treat prolonged long-term viral infections, such as hepatitis, seasonal influenza, measles, mumps, rubella, human papillomavirus, chicken pox, and herpes zoster. Sanofi and GSK plc also had a considerable market share owing to their strong portfolio and robust pipeline of candidates.
Other prominent players in the market include AstraZeneca, Johnson & Johnson, CSL SEQIRUS, Novavax, Bavarian Nordic, and others. These companies are strongly focusing on introducing technologically advanced products, such as combination vaccines, which will contribute to the market growth.
LIST OF KEY COMPANIES PROFILED:
- Merck & Co., Inc. (U.S.)
- GSK plc (U.K.)
- Sanofi (France)
- Pfizer Inc. (U.S.)
- SEQIRUS (CSL Limited) (Australia)
- Moderna, Inc. (U.S.)
- AstraZeneca (U.K.)
- Johnson & Johnson Services, Inc. (U.S.)
- Novavax (U.S.)
- Dynavax Technologies (U.S.)
KEY INDUSTRY DEVELOPMENTS:
- July 2023: GSK plc received authorization for Arexvy from the Medicines and Healthcare Products Regulatory Agency (MHRA).
- November 2022: The U.S. FDA granted a Priority Review for GSK’s Respiratory Syncytial Virus (RSV) older adult vaccine candidate.
- October 2022: Bavarian Nordic A/S entered an agreement to supply monkeypox vaccine in Latin America and the Caribbean.
- July 2022: CSL SEQIRUS entered a multi-year influenza research partnership with the University of Liverpool Pandemic Institute to advance influenza protection and pandemic preparedness.
- June 2022: Bavarian Nordic A/S entered a five-year contract with the Government of Canada to procure the IMVAMUNE smallpox vaccine.
REPORT COVERAGE
The report provides qualitative and quantitative insights into the global market and a detailed analysis of the market size and growth rate for all possible segments. Along with this, it elaborates on the market opportunities, dynamics, and competitive landscape. Various key insights presented in the report include new product launches, key industry developments, such as mergers, acquisitions, and partnerships, epidemiology: key viral diseases in key countries, pipeline analysis, and the impact of COVID-19 on the global market.
Request for Customization to gain extensive market insights.
Report Scope & Segmentation
|
ATTRIBUTE |
DETAILS |
|
Study Period |
2021-2034 |
|
Base Year |
2025 |
|
Estimated Year |
2026 |
|
Forecast Period |
2026-2034 |
|
Historical Period |
2021-2024 |
|
Growth Rate |
CAGR of 4.4% from 2026 to 2034 |
|
Unit |
Value (USD Billion) |
|
Segmentation |
By Form
|
|
By Container Format
|
|
|
By Biosafety Level Type
|
|
|
By Region
|
Frequently Asked Questions
Fortune Business Insights says that the global market size was valued at USD 59.65 billion in 2026 and is projected to reach USD 84.18 billion by 2034.
In 2025, the Asia Pacific market value stood at USD 31.43 billion.
The market will exhibit a CAGR of 4.4% during the forecast period of 2026-2034.
The liquid segment is expected to be the leading segment in this market during the forecast period.
The growing prevalence of viral infections and robust investment by market players to accelerate the development of pipeline candidates are some of the major driving factors in the global market.
Pfizer, Inc., Moderna, Inc., and Merck & Co., Inc. are some of the leading players in the global market.
Asia Pacific region dominated the market in 2025.
Get 20% Free Customization
Expand Regional and Country Coverage, Segments Analysis, Company Profiles, Competitive Benchmarking, and End-user Insights.
Related Reports
-
US +1 833 909 2966 ( Toll Free )
-
Get In Touch With Us