Hedgehog Pathway Inhibitors Market Size, Share & Industry Analysis, By Product (Vismodegib Family, Sonidegib Family, Glasdegib Family, and Next-generation Hedgehog Pathway Inhibitors), By Disease Indication (Locally advanced BCC, Metastatic BCC, Newly diagnosed AML, and Others), By Dosage Form (Oral capsules, Oral tablets, and Others), By Age Group (Pediatric and Adult), By Type (Branded and Generic), By Distribution Channel (Hospital Pharmacies, Specialty pharmacies, Retail Pharmacies, and Online Pharmacies), and Regional Forecast, 2026-2034
Hedgehog Pathway Inhibitors Market Size and Future Outlook
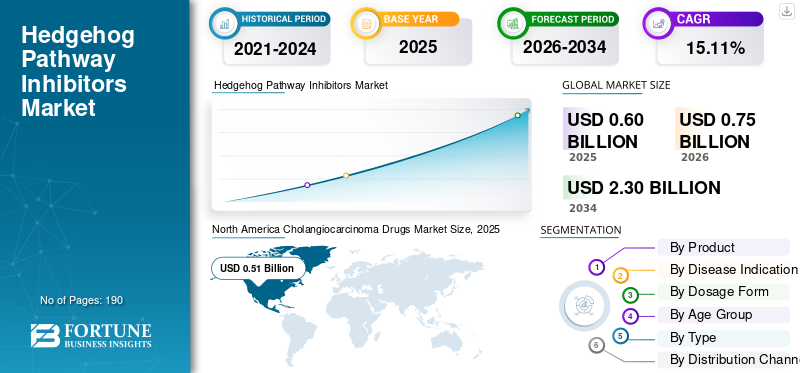
The hedgehog pathway inhibitors market size was valued at USD 0.60 billion in 2025. The market is projected to grow from USD 0.75 billion in 2026 to USD 2.30 billion by 2034, exhibiting a CAGR of 15.11% during the forecast period. North America dominated the hedgehog pathway inhibitors market with a market share of 46.66% in 2025.
The Hedgehog (Hh) pathway inhibitors offer targeted cancer therapies that block specific proteins, primarily Smoothened (SMO), in a signaling pathway that drives growth, differentiation and survival. The market is projected to grow steadily in the coming years. The growth is driven by continued use of approved hedgehog pathway-targeting drugs for advanced basal cell cancer treatments. As more patients with difficult-to-treat basal cell carcinoma require targeted treatment options when surgery or radiation are not suitable, demand for these therapies continues to rise. Additionally, the ongoing clinical development of new hedgehog pathway inhibitors is expanding the market's growth potential. Broader geographic availability of approved products and continued research into pathway-driven diseases are expected to support market expansion over the forecast period.
- For instance, in August 2025, Sol-Gel Technologies completed in its Phase 3 trial of SGT-610 (patidegib gel, 2%) for Gorlin syndrome. SGT-610 is a topical inhibitor of the hedgehog signaling pathway and, if approved, could become the first product to prevent the development of new basal cell carcinoma lesions in these patients. Such pipeline progress is expected to strengthen future treatment availability and support overall market growth.
Furthermore, leading players in the industry, such as F. Hoffmann-La Roche Ltd, Genentech, Inc, Sun Pharmaceutical Industries Ltd., and Pfizer Inc., are focusing on research and development and expanding their offerings to strengthen their market positions.
Download Free sample to learn more about this report.
HEDGEHOG PATHWAY INHIBITORS MARKET TRENDS
Pipeline Expansion Beyond Commercially Approved SMO Inhibitors is a Prominent Market Trend
A key market trend observed in the market is an increasing focus on pipeline expansion beyond the commercially approved SMO inhibitors. As more companies advance newer hedgehog pathway candidates with different formulations and broader disease targets, the market development base becomes stronger. This increases the possibility of future label expansion, improves long-term treatment availability, and reduces dependence on a small number of marketed drugs. Such continued pipeline progress is expected to strengthen investor interest, clinical attention, and future commercial growth in the market.
- For instance, in January 2025, Stamford Pharmaceuticals announced positive Phase 2 results for SP-002 in combination with 4 weeks of vismodegib in patients with multi-lesional basal cell carcinoma. The company said that the study showed positive results and highlighted the need for a non-surgical treatment option for BCC patients who are not ideal candidates for surgery. This supports the rationale that continued demand for systemic targeted treatment in difficult-to-treat BCC cases is helping drive the market growth.
MARKET DYNAMICS
MARKET DRIVERS
Download Free sample to learn more about this report.
Rising Burden of Advanced Basal Cell Carcinoma is Supporting Market Growth
The rising burden of advanced basal cell carcinoma is driving the hedgehog pathway inhibitors market growth. Basal cell carcinoma is the most common type of skin cancer, and these inhibitors are extensively used for its treatment, elevating demand as the case burden progresses to locally advanced or difficult-to-treat disease. When such patients are not suitable for surgery or radiotherapy, the need for systemic targeted treatment increases, thereby directly supporting demand for hedgehog pathway inhibitors. These factors are specifically positioned for the management of advanced basal cell carcinoma. As diagnosis levels increase and more complex cases require non-surgical treatment options, the commercial importance of this therapy class continues to strengthen.
- For instance, in May 2024, DDG published an article titled 'S2k guideline basal cell carcinoma of the skin (update 2023)' that stated that BCC is the most common malignant tumor in Central Europe and notes an incidence in Germany of at least 200 new cases per 100,000 populations every year, reinforcing the high and continuing disease burden in Europe.
MARKET RESTRAINTS
Limited Eligible Patient Population Is Restricting Market Expansion
A key factor limiting market growth is the lack of eligible patient population, as these therapies are primarily used in a narrow subset of advanced basal cell carcinoma patients. Although basal cell carcinoma is highly prevalent, most cases are treated successfully with surgery or radiotherapy that reduces the number of patients who actually require systemic hedgehog-pathway inhibitors. Additionally, the approved uses of these drugs remain tightly defined. Erivedge is indicated for metastatic BCC or locally advanced BCC in adults who are not candidates for surgery and radiation, and Odomzo is indicated for adults with locally advanced BCC after recurrence or when surgery or radiation is not appropriate. Since the class's commercial use is concentrated in this smaller, difficult-to-treat population, the overall addressable market remains limited, directly restricting broader revenue growth.
- For instance, in March 2022, the British Journal of Dermatology (BJD Journal) published an article titled 'Characterization of patients with advanced basal cell carcinoma progressing while receiving Hedgehog pathway inhibitors' that noted that while basal cell carcinoma is the most common human cancer worldwide, only a small proportion of patient’s progress to locally advanced or metastatic disease. It also highlighted that inhibitors such as vismodegib and sonidegib were developed specifically for this advanced subset, reflecting the classic commercial dependence on a narrow patient pool.
MARKET OPPORTUNITIES
Expansion into Rare Hedgehog-Pathway-Driven Disorders Creates New Market Opportunities
The market is gaining new growth opportunities through expansion into rare disorders driven by the hedgehog pathway. The expansion assisted companies move beyond the relatively narrow advanced basal cell carcinoma treatment sector. When drug developers target rare diseases associated with abnormal Hedgehog signaling, they create additional commercial opportunities in areas where treatment options are limited, and the unmet need is high. These factors also improve the long-term value of the drug class by broadening its clinical relevance outside the currently established market base. As a result, development in rare disorders can support future approvals, strengthen pipeline depth, and create incremental revenue opportunities for market. Underscoring these factors, regulatory bodies are also promoting research and development, offering reimbursement alternatives.
- For instance, in July 2025, Endeavor BioMedicines announced that taladegib (ENV-101) received Orphan Drug Designation from both the U.S. FDA and the European Commission for the treatment of Idiopathic Pulmonary Fibrosis (IPF). Taladegib is a Hedgehog signaling pathway inhibitor, showcasing that companies are actively exploring the pathway beyond traditional oncology use into rare, high-unmet-need disorders. Such expansion creates a significant opportunity for the market by opening new disease segments and increasing future development potential.
MARKET CHALLENGES
Monitoring Burden and Tolerability Issues Challenges Long-Term Market Growth
The key challenge faced by the market is monitoring burden and tolerability issues. These therapies often require close management of adverse effects during treatment. Approved products in this class are associated with side effects such as muscle spasms, alopecia, dysgeusia, weight loss, fatigue, nausea, and diarrhea. Simultaneously, sonidegib also carries precautions linked to creatine kinase elevation and related monitoring needs. Since these drugs are often used over extended periods in advanced basal cell carcinoma, persistent side effects can lead to dose interruptions, treatment discontinuation, and reduced patient adherence. This challenges the market, as strong clinical activity alone may not translate into sustained long-term use when treatment management becomes difficult in real-world settings.
- For instance, in May 2024, JAAD Journal published an article titled 'Twelve-year continuous therapy with vismodegib for advanced basal cell carcinoma: Tolerability and partial reversibility of adverse events' that highlighted that in long-term vismodegib treatment for advanced basal cell carcinoma, management of side effects becomes crucial as many patients require repeated drug holiday interruptions due to adverse events. This reflects how monitoring burden and side-effect management can directly challenge treatment continuity and restrain wider market expansion.
Segmentation Analysis
By Product
High Physician Awareness for Vismodegib Family Leads to Segmental Growth
Based on the product, the market is categorized into vismodegib family, sonidegib family, glasdegib family, and next-generation hedgehog pathway inhibitors.
Among these, the vismodegib family is estimated to dominate the market. The vismodegib family was the first commercially established hedgehog pathway inhibitor that built an early lead in physician awareness, regulatory acceptance, and real-world use in advanced basal cell carcinoma. It has created a stronger commercial base and is one of the most recognized treatment options for patients who are not suitable for surgery or radiation. Additionally, ongoing research and development initiatives are also driving the segment's growth.
- For instance, in January 2025, Stamford Pharmaceuticals announced positive Phase 2 results for SP-002 in combination with vismodegib in patients with multi-lesional basal cell carcinoma. The company stated that the study evaluated SP-002 in combination with vismodegib, a Hedgehog pathway inhibitor, and reported positive results. This shows that vismodegib remains clinically relevant in ongoing development, supporting its dominant market position.
The next-generation hedgehog pathway inhibitors segment is expected to grow at a CAGR of 42.94% over the forecast period.
To know how our report can help streamline your business, Speak to Analyst
By Disease Indication
High Utilization of Hedgehog Pathway Inhibitor for Locally Advanced BCC to Boost Segmental Growth
Based on disease indication, the market is segmented into locally advanced BCC, metastatic BCC, newly diagnosed AML, and others.
In 2025, the locally advanced BCC segment accounted for the largest share of the market by disease indication. Hedgehog pathway inhibitors are most commonly used in this setting, where patients often cannot be effectively treated with surgery or radiotherapy. This creates a direct and recurring need for systemic targeted treatment, making locally advanced BCC dominant within the market. The increased focus of key companies on expanding access and reimbursement alternatives for the disease underscores its importance.
- For instance, in July 2025, Endeavor BioMedicines announced that taladegib (ENV-101) received Orphan Drug Designation from both the U.S. FDA and the European Commission. taladegib, a Hedgehog signaling pathway inhibitor, shows that companies continue to expand investment in Hedgehog pathway-driven diseases. This supports the broader commercial importance of severe and difficult-to-treat disease settings, including locally advanced BCC, which remains the core indication base of the market.
The others segment is projected to grow at a CAGR of 27.09% during the market forecast period.
By Dosage Form
Growing Demand and Adoption of Oral Capsules Form Leads to Segment Growth
Based on the dosage form, the market is segmented into oral capsules, oral tablets, and others.
In 2025, Oral capsules dominated the market. The segment dominated as leading commercial hedgehog pathway inhibitors used in advanced basal cell carcinoma have been launched in this form, making it the most established and widely adopted format. Since oral capsules offer convenient outpatient administration and reduce dependence on infusion-based care settings, they support easier long-term treatment use in eligible patients. These factors have helped oral capsules build the large base.
- For instance, in August 2025, Sol-Gel Technologies announced that it was conducting a Phase 3 clinical trial of SGT-610 (patidegib gel, 2%) for Gorlin syndrome. The company's update is useful here as it highlights the development of a topical hedgehog inhibitor, underscoring that the market remains largely centered on established oral hedgehog products. This makes it a good recent comparison point supporting the dominance of oral capsule therapies.
In addition, others segment is projected to grow at a CAGR of 41.91% during the study period.
By Age Group
Large Adult Patient Pool in Basal Cell Carcinoma to Drive Segmental Growth
Based on age group, the market is segmented into pediatric and adult.
By age group, the adult segment accounted for the largest share of the global market. Currently approved hedgehog pathway inhibitors are primarily indicated for adults, which keeps the commercial base concentrated in this age group. Advanced basal cell carcinoma and newly diagnosed AML are also more commonly treated in adult and older adult populations, which further increases the segment growth.
- For instance, in September 2025, Endeavor BioMedicines announced that data from its Phase 2a trial of taladegib (ENV-101) in idiopathic pulmonary fibrosis had been selected for an ALERT session at the ERS Congress 2025. The release discusses clinical development in adult disease population and identifies taladegib as a hedgehog pathway inhibitor. It supports the point that active development and commercialization in this class remain strongly focused on adult patients.
Pediatric segment is projected to grow at a CAGR of 24.68% during the study period.
By Type
Higher Revenue Generation Potential of Branded Products to Boost Segment Growth
Based on type, the market is segmented into branded and generic products.
Based on type, branded drugs dominated the market. Hedgehog pathway inhibitors are used in niche oncology settings with relatively low product depth. Branded innovators continue to control physician awareness, pricing, and access. This keeps the market commercially concentrated around branded assets rather than broad generic competition. For instance, in January 2023, Sol-Gel Technologies acquired patidegib, a Phase 3, breakthrough-designated orphan product candidate, to pursue the prevention of new basal cell carcinomas in Gorlin syndrome. This shows that new value creation in the market is still centered on branded innovation rather than generic expansion.
In addition, the generic segment is projected to grow at a CAGR of 23.54% during the study period.
By Distribution Channel
Need for Specialty Follow-Up While Purchase Leads to Specialty Pharmacies Segment Growth
Based on distribution channel, the market is segmented into hospital pharmacies, specialty pharmacies, retail pharmacies, and online pharmacies.
The specialty pharmacies segment is estimated to hold the largest hedgehog pathway inhibitors market share during the forecast period. Hedgehog pathway inhibitors are niche oncology products that require controlled dispensing, patient counseling, adherence support, and monitoring for significant safety risks. Since these therapies are used in advanced or difficult-to-treat disease settings specialist follow-up and may involve pregnancy warnings, tolerability management, and, they fit more naturally into specialty pharmacy channels than into standard retail distribution. This makes specialty pharmacies a more suitable and commercially important channel for the market.
The online pharmacies segment is projected to grow at a CAGR of 22.65% over the study period.
Hedgehog Pathway Inhibitors Market Regional Outlook
By geography, the market is categorized into Europe, North America, Asia Pacific, Latin America, and Middle East & Africa.
North America
North America Cholangiocarcinoma Drugs Market Size, 2025 (USD Billion)
To get more information on the regional analysis of this market, Download Free sample
North America held a dominant share in 2024 at USD 0.22 billion and maintained its leading position in 2025 at USD 0.28 billion. The market is growing in North America as the region has a high diagnosed burden of non-melanoma skin cancer and strong access to dermatology and oncology specialists, which supports the identification and treatment of advanced BCC cases. The established availability of approved hedgehog inhibitors in the U.S. for advanced disease also supports growth.
U.S. Hedgehog Pathway Inhibitors Market
Given North America's substantial contribution, the U.S. market is estimated at around USD 0.32 billion in 2026, accounting for roughly 42.38% of global market.
Europe
Europe is projected to grow at 13.67% over the coming years, the second-highest among all regions, and reach a valuation of USD 0.20 billion by 2026. The market is growing due to supportive regulatory frameworks and mature oncology care systems across major European countries, which support treatment uptake.
U.K. Hedgehog Pathway Inhibitors Market
The U.K. market is estimated at around USD 0.04 billion in 2026, representing roughly 4.83% of the global market.
Germany Hedgehog Pathway Inhibitors Market
Germany's market is projected to reach approximately USD 0.04 billion in 2026, equivalent to around 5.91% of the global market.
Asia Pacific
Asia Pacific is estimated to reach USD 0.14 billion in 2026 and secure the position of the third-largest region in the market. The market is growing in the Asia Pacific as the region includes countries such as Australia, with a very high skin cancer burden, which increases diagnosis and treatment demand for difficult-to-treat BCC cases. Also, improving patient care and oncology infrastructure for wider access to targeted therapies across developed different countries are supporting regional growth.
Japan Hedgehog Pathway Inhibitors Market
The Japanese market in 2026 is estimated at around USD 0.04 billion, accounting for approximately 5.04% of the global market.
China Hedgehog Pathway Inhibitors Market
China's market is projected to be one of the largest worldwide, with 2026 revenues estimated at around USD 0.40 billion, representing approximately 4.72% of global sales.
India Hedgehog Pathway Inhibitors Market
The Indian market in 2026 is estimated at around USD 0.01 billion, accounting for roughly 1.69% of global revenue.
Latin America and the Middle East & Africa
The Latin America and Middle East & Africa regions are expected to witness moderate growth in this market during the forecast period. The Latin America market is set to reach a valuation of USD 0.03 billion in 2026. The market is growing in Latin America as the region sees a broader rise in cancer burden. Similarly, countries such as Brazil continue to strengthen cancer surveillance and access to specialist treatment. In the Middle East & Africa, the GCC is set to reach USD 0.02 billion in 2026.
South Africa Hedgehog Pathway Inhibitors Market
The South African market is projected to reach approximately USD 0.004 billion by 2026, accounting for roughly 0.56% of global revenue.
COMPETITIVE LANDSCAPE
Key Industry Players
Emphasis on Research and Development by Key Players to Propel Market Competition
The global market is highly consolidated, with companies such as F. Hoffmann-La Roche Ltd, Genentech, Inc, Sun Pharmaceutical Industries Ltd., Pfizer Inc., Sol-Gel Technologies Ltd., Endeavor BioMedicines, and AstraZeneca holding significant market share. Strategic partnerships, new product launches, pipeline development, and increased investments in the sector drive these companies' market share gains.
- For instance, in September 2025, Endeavor BioMedicines announced data from a Phase 2a trial evaluating the safety and efficacy of taladegib (ENV-101), a Hedgehog signaling pathway inhibitor in patients with idiopathic pulmonary fibrosis (IPF), have been selected for an ALERT (Abstracts Leading to Evolution in Respiratory Medicine Trials) session at the ERS Congress 2025. The data highlighted high-impact clinical trials in respiratory medicine.
Other notable players in the global market include BridgeBio Pharma, Inc., Curis, Inc., and Novartis AG. These companies are expected to prioritize technological advancements, strategic collaborations, and new product launches to strengthen their positions during the forecast period.
LIST OF KEY HEDGEHOG PATHWAY INHIBITORS COMPANIES PROFILED
- Hoffmann-La Roche Ltd (Switzerland)
- Genentech, Inc (U.S.)
- Sun Pharmaceutical Industries Ltd. (India)
- Pfizer Inc. (U.S.)
- Sol-Gel Technologies Ltd. (Israel)
- BridgeBio Pharma, Inc. (U.S.)
- Endeavor BioMedicines (U.S.)
- Curis, Inc. (U.S.)
- Novartis AG. (Switzerland)
- Endeavor BioMedicines (U.S.)
KEY INDUSTRY DEVELOPMENTS
- November 2025: Endeavor BioMedicines received PRIority MEdicines (PRIME) designation from the European Medicines Agency (EMA) for its investigational therapy, taladegib (ENV-101), an investigational Hedgehog signaling pathway inhibitor, for the treatment of idiopathic pulmonary fibrosis (IPF).
- October 2025: Regeneron Pharmaceuticals, Inc. announced new and updated data from its advancing oncology pipeline at the European Society for Medical Oncology (ESMO) 2025 Meeting, including new Phase 3 C-POST data on an every 6-week dosing regimen for the PD-1 inhibitor Libtayo (cemiplimab) as adjuvant treatment for cutaneous squamous cell carcinoma (CSCC) with a high risk of recurrence.
- January 2025: Stamford Pharmaceuticals Inc announced positive results from its ASN-002-003 multi-lesional clinical trial evaluating SP-002, an Adenovirus-5 replication-deficient vector encoding human Interferon, in combination with vismodegib (a Hedgehog Pathway Inhibitor) in subjects presenting with multiple BCCs.
- April 2024: Stamford Pharmaceuticals Inc initiated its locally advanced basal cell carcinoma (laBCC) trial. The study evaluated SP-002, an adenoviral-based immunotherapy, in combination with Roche's vismodegib (Erivedge), a Hedgehog Pathway inhibitor (HHPI), approved for the treatment of adult patients presenting with locally advanced and metastatic basal cell carcinoma.
- April 2024: Endeavor BioMedicines, Inc., a clinical-stage biotechnology company, received USD 132.5 million Series C financing. The financing will be used to advance the clinical development of ENV-101, its lead candidate for the treatment of idiopathic pulmonary fibrosis (IPF) and progressive pulmonary fibrosis (PPF); and to advance ENV-501, a human epidermal growth factor 3 (HER3) antibody-drug conjugate (ADC) for the treatment of HER3-positive solid tumors through clinical proof-of-concept studies.
- August 2021: IMPACT Therapeutics received approval from the National Medical Products Administration (NMPA) for clinical trials of its hedgehog inhibitor IMP5471. This will be a single-arm, open-label, multicenter, Phase I clinical study. The study aimed to evaluate the safety, tolerability, pharmacokinetics, and preliminary efficacy of IMP5471 as monotherapy or in combination with LDAC in patients with myeloid malignancies.
- January 2021: Endeavor Launches a Hedgehog Inhibitor in collaboration with Eli Lilly Company with an investment of USD 62.0 million.
REPORT COVERAGE
The global hedgehog pathway inhibitors market analysis provides a detailed study of a niche but steadily growing targeted oncology segment. It examines how the market is developing through the continued use of approved hedgehog pathway inhibitors in advanced basal cell carcinoma and the ongoing progress of next-generation pipeline candidates. The study covers key market drivers, restraints, challenges, and growth opportunities influencing future expansion across major regions. It also evaluates market size outlook, competitive positioning, regulatory developments, and recent company activities shaping the industry. In addition, the report offers segment-wise analysis by product, disease indication, dosage form, age group, type, distribution channel, and end user, helping identify the areas expected to generate the strongest commercial demand.
Request for Customization to gain extensive market insights.
Report Scope & Segmentation
| ATTRIBUTE | DETAILS |
| Study Period | 2021-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2021-2024 |
| Growth Rate | CAGR of 15.11% from 2026 to 2034 |
| Unit | Value (USD Billion) |
| Segmentation | By Product, Disease Indication, Dosage Form, Age Group, Type, Distribution Channel, and Region |
| By Product |
|
| By Disease Indication |
|
| By Dosage Form |
|
| By Age Group |
|
| By Type |
|
| By Distribution Channel |
|
| By Region |
|
Frequently Asked Questions
According to Fortune Business Insights, the global market value stood at USD 0.60 billion in 2025 and is projected to reach USD 2.30 billion by 2034.
In 2025, the North America market value stood at USD 0.28 billion.
The market is expected to grow at a CAGR of 15.11% over the forecast period of 2026-2034.
The vismodegib family segment is expected to lead the market.
Rising burden of advanced basal cell carcinoma is driving market growth.
F. Hoffmann-La Roche Ltd, Genentech, Inc, Sun Pharmaceutical Industries Ltd., Pfizer Inc., and Sol-Gel Technologies Ltd. are the major market players in the global market.
North America dominated the market in 2025.
Get 20% Free Customization
Expand Regional and Country Coverage, Segments Analysis, Company Profiles, Competitive Benchmarking, and End-user Insights.
Related Reports
-
US +1 833 909 2966 ( Toll Free )
-
Get In Touch With Us