Microbiome Therapeutics Market Size, Share & Industry Analysis, By Type (Transplantation and Drugs), By Disease Indication (Clostridium Difficile Infection (CDI), Transplant / Oncology-Support Settings, Vaginal Health, Dermatology, Inflammatory Bowel Disease, and Others), By Age Group (Adult and Pediatrics), By Route of Administration (Rectal, Oral, Topical, and Others), By Distribution Channel (Hospital Pharmacies, Specialty Pharmacies, and Others), and Regional Forecast, 2026-2034
KEY MARKET INSIGHTS
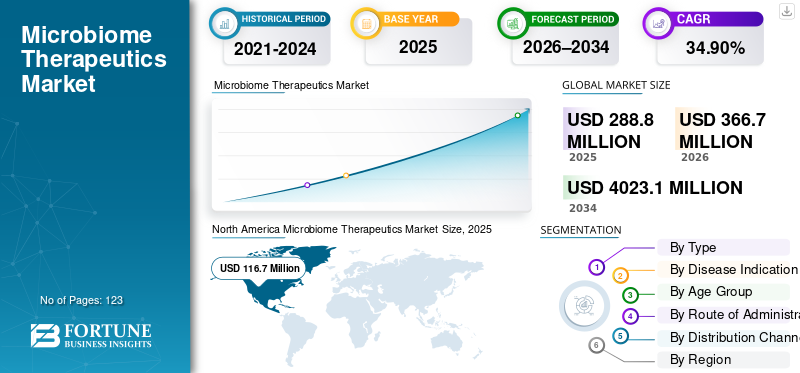
The global microbiome therapeutics market size was valued at USD 288.8 million in 2025. The market is projected to grow from USD 366.7 million in 2026 to USD 4,023.1 million by 2034, exhibiting a CAGR of 34.90% during the forecast period. North America dominated the microbiome therapeutics market with a market share of 40.40% in 2025.
The global microbiome therapeutics market is anticipated to witness accelerating growth in the forthcoming years. These microbiome therapies use probiotics, fecal microbiota transplants (FMT), prebiotics, or engineered microbes to treat conditions such as C. difficile infection, IBD, and metabolic disorders. Advancements in microbiome sequencing capabilities, rising research initiatives, and the increasing prevalence of gut infections are collectively driving market growth.
Emphasizing these key drivers, many companies are directing their resources toward research and development, attracting further investment to strengthen their market position.
- For instance, in September 2025, Metagen Therapeutics, Inc., a biotech company advancing medicine and drug discovery based on the human gut microbiome, raised USD 15.0 million in a Series B financing round. Such developments are anticipated to boost the overall market growth.
Furthermore, leading players in the microbiome therapeutics industry, such as Seres Therapeutics, Finch Therapeutics Group, Inc., Ferring, and MaaT Pharma, are directing their resources toward research and development, expanding their offerings, and strengthening their market positions.
Download Free sample to learn more about this report.
MICROBIOME THERAPEUTICS MARKET TRENDS
Shift Towards Engineered and Targeted Modalities to Provide Precision Interventions Is a Market Trend
A major global trend in the microbiome therapeutics market observed is the shift towards engineered and targeted modalities to deliver precision interventions. Targeted microbiome modalities, such as engineered bacteriophages and strain-targeted LBPs, are gaining popularity. These novelties are anticipated to overcome challenges with earlier microbiome therapeutic approaches, such as inconsistent results and difficulty in controlling them. This precision also supports differentiation and pricing power, offering purpose-built treatment. As antibiotic resistance and recurrence remain costly, targeted solutions paired with diagnostics create a more predictable adoption path and accelerate pipeline activity.
- For instance, in November 2024, Microbiotica, a company developing a pipeline of precision microbiome medicines called live biotherapeutic products (LBPs), dosed its first patient in its Phase 1b First-in-Human trial, COMPOSER-1, of MB310 in patients with ulcerative colitis (UC). Such developments are expected to boost market growth.
Download Free sample to learn more about this report.
MARKET DYNAMICS
MARKET DRIVERS
Expanding Therapeutic Range of Microbiomes to Fuel the Market Growth
One of the key factors driving the global microbiome therapeutics market growth is the expanding indications beyond recurrent C. difficile into oncology, metabolic disorders, and inflammatory diseases. The expanded breadth of applications of microbiome therapeutics increases the patient pool and the clinical settings in which they are administered. These factors encourage greater pipeline investment, strategic partnerships, and increased funding activities. Additionally, adding chronic diseases further increases the addressable market, translating into higher lifetime value per patient if clinical benefit is proven. Such advantages attract co-development deals and accelerate adoption pathways.
- For instance, in January 2026, EnteroBiotix Limited, a company focused on developing therapies for gut health, showcased positive final results from TrIuMPH, a Phase 2a clinical trial evaluating EBX-102-02, an oral full-spectrum microbiome therapeutic, in patients with irritable bowel syndrome with constipation (IBS-C) or diarrhoea (IBS-D)—such developments are expected to drive market growth.
MARKET RESTRAINTS
Regulatory Uncertainty and Strict Regulations to Hamper Market Development
Regulatory uncertainty is a significant market restraint for microbiome therapeutics. These products are living, complex mixtures, so regulators often refine expectations for safety and tolerance. Strict regulations are required for the identification of strains, their range, potency, and long-term clinical benefit. Companies may need to repeat analytics work or adjust trial protocols in response to evolving regulations. Such factors create approval risk, longer timelines, and higher costs, which can delay launches and make pharmaceutical companies more cautious, slowing overall market growth.
- For instance, in August 2025, BiomX disclosed that the U.S. FDA placed a clinical hold on the U.S. portion of its BX004 Phase 2b trial (cystic fibrosis) for a phage therapy targeting specific pathogenic bacteria and requested additional data. Such developments slow down pharmaceutical development and limit the market's growth potential.
MARKET OPPORTUNITIES
Development of Oral Formulations to Offer Market Growth Opportunity
A key market growth opportunity in microbiome therapeutics is to increase funding and investment, which will propel growth. A rise in investment facilitates clinical trials and builds GMP manufacturing capacity for live Biotherapeutics. Such developments also speed up market access planning and enable leading companies to scale their businesses. The net effect is a more stable competitive landscape with better-funded leaders and faster translation of pipelines into approved therapies, expanding overall market growth potential.
- For instance, in September 2025, MRM Health NV, a biopharmaceutical company focused on microbiome-based therapeutics for inflammatory diseases and immune-oncology, received USD 64.0 million in funding to support a Phase 2b trial of the company’s program MH002 in ulcerative colitis. Such developments offer market growth opportunities.
MARKET CHALLENGES
High Cost of Microbiome Therapeutics Poses a Critical Challenge to Market Growth
The overall microbiome therapeutics market faces a critical challenge: inconsistent responses. Patient outcomes vary widely based on numerous factors, including prior antibiotic exposure, diet, disease severity, concurrent medications, and the patient’s baseline microbiome. When the same therapy performs well in some patients but not others, trials can yield a weaker average benefit, making it harder to hit primary endpoints and build a clear value story for doctors and payers. This uncertainty increases development risk, pushes companies to run larger and more expensive studies, thereby restraining overall market growth.
- For instance, in August 2025, Vedanta Biosciences announced that its oral microbiome candidate, VE202, did not meet the primary endpoint in the Phase 2 COLLECTiVE202 study in patients with mild-to-moderate ulcerative colitis due to inconsistent clinical response. Such factors result in setbacks in microbiome therapeutics.
Segmentation Analysis
By Type
High Usage of Microbiome Transplantation to Lead the Segmental Growth
Based on type, the market is categorized into transplantation and drugs.
Among these, the transplantation segment accounted for the largest share of the microbiome therapeutics market in 2025. The segment held a significant share because it offered a comprehensive, immediate way to restore a damaged gut ecosystem. When patients with severe dysbiosis after antibiotics require rapid microbiome recovery, transplantation-based approaches can deliver a broad, functional microbial community in a single intervention, increasing clinician confidence and accelerating adoption in real-world practice. Due to these advantages, healthcare providers have been increasingly preferring transplantation microbiome therapeutics, leading to the segment's growth.
Furthermore, strategic collaborations among key companies and the launch of new products offering innovative solutions support market growth.
- For instance, in July 2025, Clinigen partnered with MaaT Pharma for exclusive licensing and distribution, as well as commercial supply and future access, of the microbiota-based therapy (MaaT013) in Europe and the U.K. Such developments are expected to drive the segment's growth.
The microbiome drugs segment is expected to grow at a CAGR of 72.83% over the forecast period.
To know how our report can help streamline your business, Speak to Analyst
By Disease Indication
High Incidence Rate of Clostridium difficile infection (CDI) to Lead Segmental Growth
Based on the disease indication, the market is segmented into Clostridium difficile infection (CDI), transplant/ oncology support settings, vaginal health, dermatology, inflammatory bowel disease, and others.
In 2025, Clostridium difficile infection (CDI) dominated the microbiome therapeutics market share due to increasing incidence and a high recurrence rate. The disease causes high microbiome disruption and leads to high spending. These factors have led pharmaceutical companies to increasingly focus on new pipeline candidates and their regulatory approvals, leading to growth in the segment.
- For instance, in January 2024, Seres Therapeutics, Inc. received Fast Track Designation from the U.S. FDA for SER-155 to prevent the recurrence of Clostridioides difficile infection (CDI) in adults following antibacterial treatment for recurrent CDI (rCDI).
The dermatology segment is projected to grow at a CAGR of 53.46% during the forecast period.
By Age Group
Presence of Approved Products for Adults to Drive Segmental Growth
Based on age group, the market is segmented into adult and pediatric.
In 2025, the adult segment dominated the market due to the high recurrence of CDI in adults. Additionally, many transplant and oncology support use cases are common and are actively addressed in adult care pathways. Additionally, the presence of approved products for microbiome therapeutics in adults reinforces the segment's dominance.
- For instance, in April 2023, Seres Therapeutics, Inc. received approval from the U.S. FDA for VOWSTTM, an orally administered microbiota-based therapeutic to prevent recurrence of C. difficile Infection (CDI) in adults following antibacterial treatment for recurrent CDI (rCDI).
In addition, the pediatric segment is projected to grow at a CAGR of 46.50% during the study period.
By Route of Administration
Direct Delivery of Rectal Therapy to Reinforce Their Dominance in The Market
Based on route of administration, the market is segmented into rectal, oral, topical, and others.
In 2025, the rectal delivery dominated because it enables direct, localized delivery to the gut with a predictable route under clinician supervision. patients who are at high risk and outcomes need to be tightly managed. Underscoring these advantages, many key companies are also focusing on new product launches in the segment.
- For instance, in November 2022, Ferring Pharmaceuticals received approval from the U.S. FDA for REBYOTA (fecal microbiota, live-jslm), indicated for the prevention of recurrence of Clostridioides difficile infection (CDI) in individuals 18 years of age and older following antibiotic treatment for recurrent CDI.
In addition, the oral segment is projected to grow at a CAGR of 48.66% during the forecast period.
By Distribution Channel
High Utilization of Microbiome Therapeutics by Healthcare Providers Leads Hospital Pharmacies Segmental Growth
Based on the distribution channel, the market is segmented into hospitals, pharmacies, specialty pharmacies, and others.
The hospital pharmacies held a significant market share in the global market for 2025. The large market share is attributed to the segment, as these microbiome therapeutics are administered in controlled settings under clinical guidance. Hospital pharmacies supply these critical medicines. Hospitals also have a direct incentive to adopt them through inpatient and hospital-connected pathways, thereby lifting hospital pharmacies' share.
- For instance, in October 2025, Ferring announced real-world analyses for REBYOTA presented at IDWeek 2025, highlighting effectiveness and routine-practice use—an adoption pattern that typically runs through hospital-led infectious disease care and hospital pharmacy
The specialty pharmacies and drug stores segment is projected to grow at a CAGR of 55.57% during the study period.
Microbiome Therapeutics Market Regional Outlook
By region, the market is categorized into Europe, North America, Asia Pacific, Latin America, and the Middle East & Africa.
North America
North America Microbiome Therapeutics Market Size, 2025 (USD Million)
To get more information on the regional analysis of this market, Download Free sample
North America held the dominant share in 2024, valued at USD 92.3 million, and maintained its leading position in 2025, with a value of USD 116.7 million. The market in North America is expected to grow significantly over the forecast period, driven by new product launches, expanding pipeline, and increasing investment in the region. These factors are facilitating market growth.
U.S. Microbiome Therapeutics Market
Given North America’s strong contribution and the U.S. dominance in the region, the U.S. market can be estimated at around USD 136.8 million in 2026, accounting for roughly 37.31% of the global market.
Europe
Europe is projected to grow at 35.20% over the coming years, the second-highest among all regions, and reach a valuation of USD 89.4 million by 2026. The area is expected to experience robust growth driven by the increasing prevalence of infectious diseases and the rising geriatric population.
U.K. Microbiome Therapeutics Market
The U.K. market in 2026 is estimated at around USD 13.9 million, representing roughly 3.78% of the global market.
Germany Microbiome Therapeutics Market
Germany’s market is projected to reach approximately USD 23.1 million in 2026, equivalent to around 6.31% of the global market.
Asia Pacific
Asia Pacific is estimated to reach USD 73.2 million in 2026 and secure the position of the third-largest region in the market.
Japan Microbiome Therapeutics Market
The Japanese market in 2026 is estimated to be around USD 17.8 million, accounting for approximately 4.84% of the global market.
China Microbiome Therapeutics Market
China’s market is projected to be one of the largest worldwide, with 2026 revenues estimated at around USD 24.6 million, representing approximately 6.71% of global sales.
India Microbiome Therapeutics Market
The Indian market in 2026 is estimated at around USD 6.0 million, accounting for roughly 1.64% of global revenue.
Latin America and the Middle East & Africa
The Latin America and Middle East & Africa regions are expected to witness moderate growth in this market space during the forecast period. The Latin America market is set to reach a valuation of USD 15.4 million in 2026. The region is experiencing market growth driven by increased government support. In the Middle East & Africa, the GCC is set to reach USD 19.5 million in 2026.
South Africa Microbiome Therapeutics Market
The South African market is projected to reach approximately USD 6.0 million by 2026, accounting for roughly 1.65% of the global revenue.
COMPETITIVE LANDSCAPE
Key Industry Players
Focus on New Product Launches by Key Players to Propel Market Progress
The global microbiome therapeutics market is highly consolidated, with companies such as Seres Therapeutics, Finch Therapeutics Group, Inc., and Ferring. MaaT Pharma. holding a significant market share. Strategic partnerships, expanding pipelines, technological advancements, and increased investments in the sector drive these companies' market share.
- For instance, in July 2025, MaaT Pharma announced an exclusive commercialization partnership with Clinigen for Xervyteg in acute graft-versus-host disease in Europe —such developments aimed to drive market growth.
Other notable players in the global market include Vedanta Biosciences, Inc., and Evelo Biosciences. These companies are expected to prioritize technological advancements, strategic collaborations, and new product launches to strengthen their position in the market.
LIST OF KEY MICROBIOME THERAPEUTICS COMPANIES PROFILED
- Seres Therapeutics (U.S.)
- Finch Therapeutics Group, Inc. (U.S.)
- Ferring (Switzerland)
- MaaT Pharma (France)
- Vedanta Biosciences, Inc. (U.S.)
- Nestlé Health Science (Switzerland)
- Evelo Biosciences (U.S.)
- Enterome (France)
- Synlogic (U.S.)
KEY INDUSTRY DEVELOPMENTS
- May 2025: Ferring showcased initial findings from the investigational Phase 3b multi-center, single-arm CDI-SCOPE study evaluating the safety and effectiveness of REBYOTA (fecal microbiota, live – jslm) when administered by colonoscopy.
- June 2024: Nestlé Health Science acquired global rights to Vowst from Seres Therapeutics, Inc. The transaction allowed the company to have complete control over the further development, commercialization, and manufacturing of VOWST in the US and worldwide.
- September 2024: Nexilico, a pioneer in precision microbiome engineering, collaborated with Siolta Therapeutics, a leader in the development of live biotherapeutic products (LBPs to address necrotizing enterocolitis (NEC) and develop targeted Biotherapeutics, setting the stage for innovative treatment approaches in infant health.
- September 2024: The European Microbiome Innovation for Health (EMIH) association collaborated with the Microbiome Therapeutics Innovation Group (MTIG) and EMIH to advance microbiome drug development.
- October 2023: Kanvas Biosciences acquired two active therapeutics programs, a microbial library, and intellectual property from Federation Bio, a manufacturer of live biotherapeutics products (LBPs).
REPORT COVERAGE
Request for Customization to gain extensive market insights.
Report Scope & Segmentation
|
ATTRIBUTE |
DETAILS |
|
Study Period |
2021-2034 |
|
Base Year |
2025 |
|
Estimated Year |
2026 |
|
Forecast Period |
2026-2034 |
|
Historical Period |
2021-2024 |
|
Growth Rate |
CAGR of 34.90% from 2026-2034 |
|
Unit |
Value (USD Million) |
|
Segmentation |
By Type, Disease Indication, Age Group, Route of Administration, Distribution Channel, and Region |
|
By Type |
· Transplantation · Drugs |
|
By Disease Indication |
· Clostridium difficile infection (CDI) · Transplant / oncology-support settings · Vaginal Health · Dermatology · Inflammatory Bowel Disease · Others |
|
By Age Group |
· Adult · Pediatric |
|
By Route of Administration |
· Rectal · Oral · Topical · Others |
|
By Distribution Channel |
· Hospital Pharmacies · Specialty Pharmacies · Others |
|
By Region |
· North America (By Type, Disease Indication, Age Group, Route of Administration, Distribution Channel, and Country) o U.S. o Canada · Europe (By Type, Disease Indication, Age Group, Route of Administration, Distribution Channel, and Country/Sub-region) o Germany o U.K. o France o Spain o Italy o Scandinavia o Rest of Europe · Asia Pacific (By Type, Disease Indication, Age Group, Route of Administration, Distribution Channel, and Country/Sub-region) o China o Japan o India o Australia o Southeast Asia o Rest of Asia Pacific · Latin America (By Type, Disease Indication, Age Group, Route of Administration, Distribution Channel, and Country/Sub-region) o Brazil o Mexico o Rest of Latin America · Middle East & Africa (By Type, Disease Indication, Age Group, Route of Administration, Distribution Channel, and Country/Sub-region) o GCC o South Africa o Rest of Middle East & Africa |
Frequently Asked Questions
According to Fortune Business Insights, the global market value stood at USD 288.8 million in 2025 and is projected to reach USD 4,023.1 million by 2034.
In 2025, the market value stood at USD 116.7 million.
The market is expected to exhibit a CAGR of 34.90% during the forecast period of 2026-2034.
By type, the transplantation segment is expected to lead the market.
The regulatory approval and expanding pipeline for microbial therapeutics are driving market growth.
Seres Therapeutics, Finch Therapeutics Group, Inc., Ferring, MaaT Pharma, and Vedanta Biosciences, Inc. are the major players in the global market.
North America dominated the market in 2025.
Get 20% Free Customization
Expand Regional and Country Coverage, Segments Analysis, Company Profiles, Competitive Benchmarking, and End-user Insights.
Related Reports
-
US +1 833 909 2966 ( Toll Free )
-
Get In Touch With Us